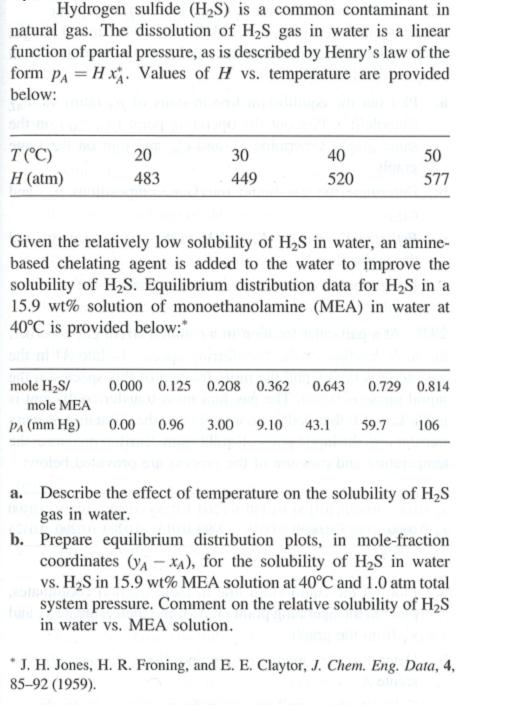
Hydrogen sulfide (HS) is a common contaminant in natural gas. The dissolution of HS gas in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959). Hydrogen sulfide (H₂S) is a common contaminant in natural gas. The dissolution of H₂S gas in water is a linear function of partial pressure, as is described by Henry's law of the form PAHx. Values of H vs. temperature are provided below: T (°C) H (atm) 20 483 30 449 mole H₂S/ 40 520 50 577 Given the relatively low solubility of H₂S in water, an amine- based chelating agent is added to the water to improve the solubility of H₂S. Equilibrium distribution data for H₂S in a 15.9 wt% solution of monoethanolamine (MEA) in water at 40°C is provided below:* 0.000 0.125 0.208 0.362 0.643 0.729 0.814 mole MEA PA (mm Hg) 0.00 0.96 3.00 9.10 43.1 59.7 106 a. Describe the effect of temperature on the solubility of H₂S gas in water. b. Prepare equilibrium distribution plots, in mole-fraction coordinates (VA-XA), for the solubility of H₂S in water vs. H₂S in 15.9 wt% MEA solution at 40°C and 1.0 atm total system pressure. Comment on the relative solubility of H₂S in water vs. MEA solution. J. H. Jones, H. R. Froning, and E. E. Claytor, J. Chem. Eng. Data, 4, 85-92 (1959).
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
The table below presents equilibrium distribution data for four gaseous solutes dissolved in water, using air as the carrier gas: a. Using a spreadsheet to perform the calculations, prepare a graph...
-
Natural gas frequently contains hydrogen sulfide, H2S. H2S is removed from natural gas by passing it through aqueous ethanolamine, HOCH2CH2NH2 (an ammonia derivative), which reacts with the hydrogen...
-
Hydrogen sulfide can be removed from natural gas by the reaction 2H2S(g) + SO2(g) 3S(s) + 2H2O(g) Calculate Go and K (at 298 K) for this reaction. Would this reaction be favored at a high or low...
-
Exercises 5-8: Sort the list of numbers from smallest to largest and display the result in a table. (a) Determine the maximum and minimum values. (b) Calculate the mean and median. Round each result...
-
A stock is currently priced at $64 and will move up by a factor or 1.18 or down by a factor of .85 each period over each of the next two periods. The risk-free rate of interest is 3 percent. What is...
-
An instructor has graded 19 exam papers submitted by students in a class of 20 students, and the average so far is 70. (The maximum possible score is 100.) How high would the score on the last paper...
-
Operating Cycle and Current Receivables} a. Dither and Sly are lawyers who specialize in income tax law. They complete their typical case in six months or less and collect from the client within one...
-
Scare-2-B-U (S2BU) specializes in costumes for all occasions. The average price of each of its costumes is $160. For each occasion, S2BU receives a 20 percent deposit two months before the occasion,...
-
QUESTION S Graph the following function on your calculator: f(x) = -0.0015x4 + 0.54x3 _ 12.6x2 - 5,280x + 288,000 Which of the following statements about f(x) is true? O This function has an absolute...
-
Logan B. Taylor is a widower whose wife, Sara, died on June 6, 2013. He lives at 4680 Dogwood Lane, Springfield, MO 65801. He is employed as a paralegal by a local law firm. For 2015, he reported the...
-
a. Wilberforce construction limited has equipment for compaction. Measure the effect of compaction on civil engineering project. b. Write an open letter to your client because the sand and timber...
-
The balance sheet shows the following: Net investment in capital assets 2015 - $243,499 2014 - $231,850 2013 - $218,268 Based upon the current cost structure for the organization, if all construction...
-
Why did the subprime crisis of 2007-2009 have such a global impact on financial markets, insititutions and the real economy? What were the differences in policies adopted in the UK and USA to...
-
You have a stock indexed portfolio worth $20 million today. The index's value today is 1,000. You enter a short position on 75 futures contracts, with the current futures price of 1,200. You close...
-
Consider the internal controls listed below. For each one, explain: ( i ) the risk it addresses, ( ii ) the risk category from Brown s taxonomy, ( iii ) the broad purpose of internal control it...
-
Laila has a portfolio of RM1 million and she found during this pandemic Covid, the Kuala Lumpur Composite Index (KLCI) has dropped by 10% since last March. She refers to you as to get your advised...
-
Which is the most accurate statement about corporations in the current digital age? Question 8 options: The lines of authority are blurry in today's information-age workplaces, and the roles of execut
-
Subtract the polynomials. (-x+x-5) - (x-x + 5)
-
Why is it important that radioisotopes used as diagnostic tools in nuclear medicine produce gamma radiation when they decay? Why are alpha emitters not used as diagnostic tools?
-
Carbon disulfide (CS2) is a toxic, highly flammable substance. The following thermodynamic data are available for CS2(l) and CS2(g) at 298 K: (a) Draw the Lewis structure of the molecule. What do you...
-
(a) Increase [NH3], increase yield NO (b) Increase [H2O], decrease yield NO (c) Decrease [O2], decrease yield NO (d) Decrease container volume, decrease yield NO (fewer moles gas in reactants) (e)...
-
Accounting for joint products You have just been hired as the chief cost accountant for the J&M Company. Its major product group is four food processing solutions, which are pro- duced in a joint...
-
Analyzing the impact of different joint costing methods Petro-Edge Corporation manu- factures specialized oils and lubricants for the aviation industry. One product group, manufactured in a joint...
-
Analyzing Joint and by-product data with standard costs Haverly Company makes two chemical solvents used in the production of cleaning compounds. The products are manufactured in a joint production...

Study smarter with the SolutionInn App


