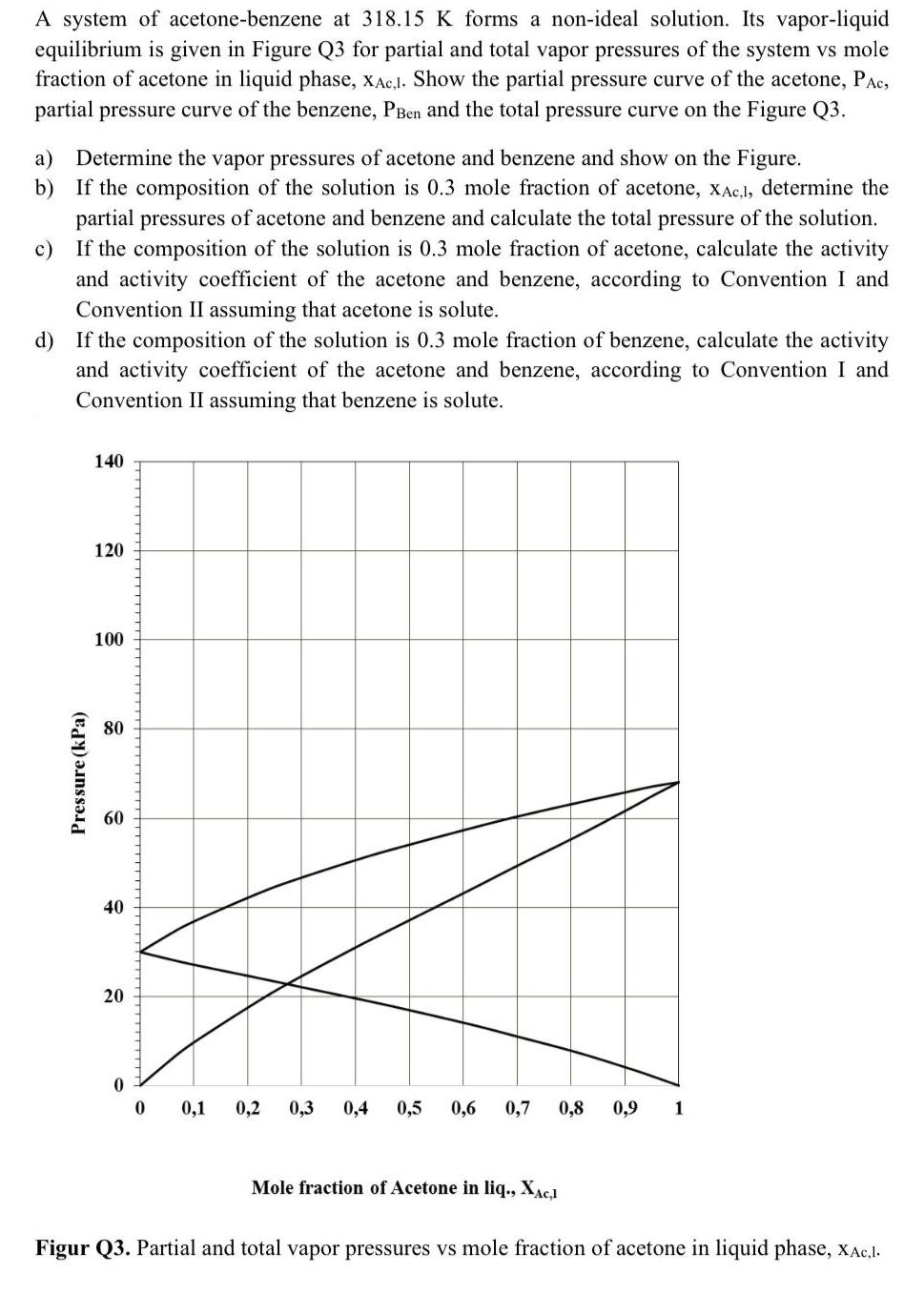
A system of acetone-benzene at 318.15 K forms a non-ideal solution. Its vapor-liquid equilibrium is given...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A system of acetone-benzene at 318.15 K forms a non-ideal solution. Its vapor-liquid equilibrium is given in Figure Q3 for partial and total vapor pressures of the system vs mole fraction of acetone in liquid phase, xAc,I. Show the partial pressure curve of the acetone, PAc, partial pressure curve of the benzene, PBen and the total pressure curve on the Figure Q3. a) Determine the vapor pressures of acetone and benzene and show on the Figure. b) If the composition of the solution is 0.3 mole fraction of acetone, xAc,l, determine the partial pressures of acetone and benzene and calculate the total pressure of the solution. If the composition of the solution is 0.3 mole fraction of acetone, calculate the activity and activity coefficient of the acetone and benzene, according to Convention I and c) Convention II assuming that acetone is solute. d) If the composition of the solution is 0.3 mole fraction of benzene, calculate the activity and activity coefficient of the acetone and benzene, according to Convention I and Convention II assuming that benzene is solute. 140 120 100 80 60 40 20 0,1 0,2 0,3 0,4 0,5 0,6 0,7 0,8 0,9 1 Mole fraction of Acetone in liq., XAC1 Figur Q3. Partial and total vapor pressures vs mole fraction of acetone in liquid phase, xAc,1. Pressure (kPa) A system of acetone-benzene at 318.15 K forms a non-ideal solution. Its vapor-liquid equilibrium is given in Figure Q3 for partial and total vapor pressures of the system vs mole fraction of acetone in liquid phase, xAc,I. Show the partial pressure curve of the acetone, PAc, partial pressure curve of the benzene, PBen and the total pressure curve on the Figure Q3. a) Determine the vapor pressures of acetone and benzene and show on the Figure. b) If the composition of the solution is 0.3 mole fraction of acetone, xAc,l, determine the partial pressures of acetone and benzene and calculate the total pressure of the solution. If the composition of the solution is 0.3 mole fraction of acetone, calculate the activity and activity coefficient of the acetone and benzene, according to Convention I and c) Convention II assuming that acetone is solute. d) If the composition of the solution is 0.3 mole fraction of benzene, calculate the activity and activity coefficient of the acetone and benzene, according to Convention I and Convention II assuming that benzene is solute. 140 120 100 80 60 40 20 0,1 0,2 0,3 0,4 0,5 0,6 0,7 0,8 0,9 1 Mole fraction of Acetone in liq., XAC1 Figur Q3. Partial and total vapor pressures vs mole fraction of acetone in liquid phase, xAc,1. Pressure (kPa)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The current in a 50-mH inductor is given in Figure P5.29. Sketch the inductor voltage. 100 :/68 10 t(ms) -100 1-- Figure P5.29
-
A Nichols chart is given in Figure E9.14 for a system with Gc(jÏ)G(jÏ)). Using the following table, find (a) the peak resonance MpÏ) in dB; (b) the resonant frequency cor; (c) the 3-dB...
-
An enthalpy-concentration diagram is given in Figure for a mixture of n-hcxanc (H). and n-octane (0) at 101 kPa. Using this diagram, determine the following: (a) The mole-fraction composition of the...
-
If you were in charge of B&D, indicate the possible decisions you might take as regards the four Ps and the points you would want to keep in mind as you go about the task of marketing for...
-
The gas mileages, rounded to the nearest number of miles per gallon, of all new car models We have presented a data scenario. In each case, decide which type of grouping (single-value, limit, or...
-
A square with side L flies past you at speed v, in a direction parallel to two of its sides. You stand in the plane of the square. When you see the square at its nearest point to you, show that it...
-
A sociologist surveys a sample of college students to determine whether there are differences in the attitudes and behaviors of male and female students. The survey contains 20 questions. For one...
-
Hitachi, Ltd., reports total revenues of 9,041,071 million for its fiscal year ending March 31, 2013, and its March 31, 2013, unadjusted trial balance reports a debit balance for trade receivables...
-
The intent of your Professional Portfolio is to serve as a tool to assist you on embarking on a successful career in the Health Informatics & Information Management Profession. The keys to success...
-
A home network is shown in the following figure and its configurations are shown in the table below. rest of Internet E Router interface D E home network WAN side IP address 130.155.169.8 IP address...
-
1. The T-beam is made from concrete having a specific weight of 150 lb/ft. Determine the dead load per foot length of the beam. Neglect the weight of the steel reinforcement. 40 in. 26 in. 10 in.- 8...
-
Use the information in the following adjusted trial balance for the Wilson Trucking Company. Account Title Cash Accounts receivable Office supplies Trucks Accumulated depreciation-Trucks Land...
-
USINT a minimum of 5 REAL PEER REVIEWED ARTICLES ~ I will be checking them. Use in-text citations and references in a.pa style. For this week's assignment, you need to create a translational study...
-
Assume a parent company owns a 100% controlling interest in its long-held subsidiary. The following excerpts are from the parent's and subsidiary's pre-consolidation income statements for the year...
-
Suppose that a wind is blowing in the direction S45E at a speed of 40 km/h. A pilot is steering a plane in the direction N60E at an airspeed (speed in still air) of 100 km/h. The true course, or...
-
1. do you agree or disagree and elaborate. QUESTION: ASLEAN WISNER. CASE STUDY 7 These are how Bill Gates possess all five types of power: 1) Reward power: Because Bill Gates is the founder of...
-
I need an essay about 1500 words about ( The structure and functions of Supreme Courts. A comparison between civil law and common law).
-
Michelles trust is subject to 3.8% surtax on the lesser of the trusts net investment income or the excess of the trusts adjusted gross income over the $12,400 threshold (the highest trust tax rate)....
-
Consider a unity negative feedback system with where K is set equal to 100 in order to achieve a specified Kv = 2. We wish to add a lead-lag compensator Show that the gain margin of the compensated...
-
Consider the system in state variable form x = Ax + Bu y = Cx + Du with (a) Compute the transfer function G(s) = Y(s)/U(s). (b) Determine the poles and zeros of the system. (c) If possible, represent...
-
The dynamics of a rocket are represented by and state variable feedback is used, where = - x1 - 25x2 + r.. Determine the roots of the characteristic equation of this system and the response of the...
-
The fly balls of spring loaded governor of Hartnell type running at 600 rpm have a radius of rotation of 80 mm with sleeve in mid-position and ball arms vertical. The ball arms and sleeve arms are of...
-
The height of Watt's governor is proportional to (a) speed (N) (b) \(\mathrm{N}^{2}\) (c) \(1 / \mathrm{N}\) (d) \(1 / \mathrm{N}^{2}\).
-
What is the main function of a governor? How does it differ from that of a flywheel?

Study smarter with the SolutionInn App


