Complete the following questions based on the remote laboratory procedures: 1. What are some similarities in...
Fantastic news! We've Found the answer you've been seeking!
Question:
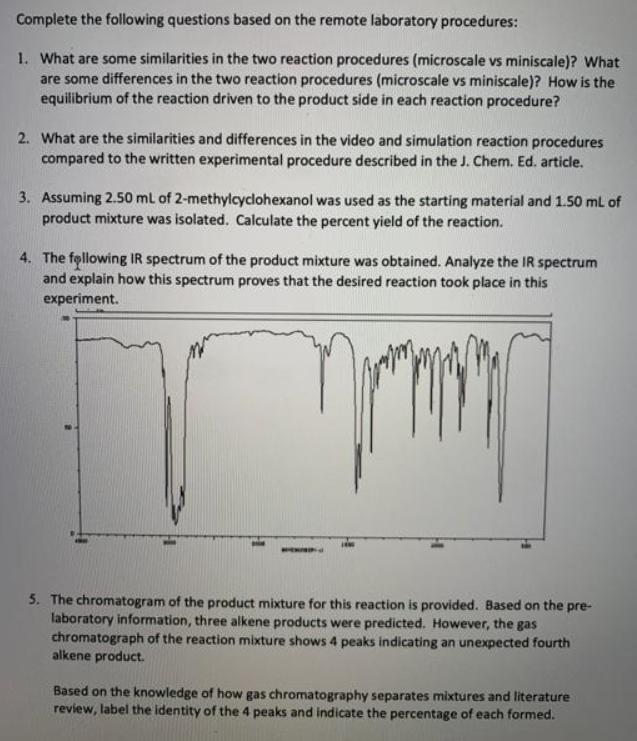
Transcribed Image Text:
Complete the following questions based on the remote laboratory procedures: 1. What are some similarities in the two reaction procedures (microscale vs miniscale)? What are some differences in the two reaction procedures (microscale vs miniscale)? How is the equilibrium of the reaction driven to the product side in each reaction procedure? 2. What are the similarities and differences in the video and simulation reaction procedures compared to the written experimental procedure described in the J. Chem. Ed. article. 3. Assuming 2.50 ml of 2-methylcyclohexanol was used as the starting material and 1.50 mL of product mixture was isolated. Calculate the percent yield of the reaction. 4. The following IR spectrum of the product mixture was obtained. Analyze the IR spectrum and explain how this spectrum proves that the desired reaction took place in this experiment. 5. The chromatogram of the product mixture for this reaction is provided. Based on the pre- laboratory information, three alkene products were predicted. However, the gas chromatograph of the reaction mixture shows 4 peaks indicating an unexpected fourth alkene product. Based on the knowledge of how gas chromatography separates mixtures and literature review, label the identity of the 4 peaks and indicate the percentage of each formed. Complete the following questions based on the remote laboratory procedures: 1. What are some similarities in the two reaction procedures (microscale vs miniscale)? What are some differences in the two reaction procedures (microscale vs miniscale)? How is the equilibrium of the reaction driven to the product side in each reaction procedure? 2. What are the similarities and differences in the video and simulation reaction procedures compared to the written experimental procedure described in the J. Chem. Ed. article. 3. Assuming 2.50 ml of 2-methylcyclohexanol was used as the starting material and 1.50 mL of product mixture was isolated. Calculate the percent yield of the reaction. 4. The following IR spectrum of the product mixture was obtained. Analyze the IR spectrum and explain how this spectrum proves that the desired reaction took place in this experiment. 5. The chromatogram of the product mixture for this reaction is provided. Based on the pre- laboratory information, three alkene products were predicted. However, the gas chromatograph of the reaction mixture shows 4 peaks indicating an unexpected fourth alkene product. Based on the knowledge of how gas chromatography separates mixtures and literature review, label the identity of the 4 peaks and indicate the percentage of each formed.
Expert Answer:
Answer rating: 100% (QA)
O Simitarities are iven below Borh reactios will do in laboratoy scale BoM reachion oeme in ytass eq... View the full answer

Related Book For 

Marketing The Core
ISBN: 978-0078028922
5th edition
Authors: Roger A. Kerin, Steven W. Hartley, William Rudelius
Posted Date:
Students also viewed these languages questions
-
What are some similarities in the world's financial systems?
-
Complete the following analogies. What are some similarities and differences between the two concepts involved in each? a. Depreciation is to plant assets as is to intangible assets . b. Depreciation...
-
What are some similarities and differences between skimming pricing, prestige pricing, and above-market pricing?
-
How many of the following could be valid JDBC URL formats for an imaginary driver named magic and a database named box? A. Only first B. Only second C. Only third D. first and second E. first and...
-
What is meant by antecedent conditions? How do they shape the value of objects?
-
Branding distinguishes one company's goods or services from those of its competitors. Each company you purchase from hopes that you will become loyal to its brand. Some well-known brands are Amazon,...
-
A 6 cylinder, 4 stroke I.C. engine is to develop \(89.5 \mathrm{~kW}\) at \(800 \mathrm{rpm}\). The stroke to bore ratio is 1.25:1. Assuming mechanical efficiency of \(80 \%\) and brake mean...
-
The following is a list of selected accounts and ending account balances taken from the books of the Adams Company on December 31, 2007: Account Title Amount Premium on preferred stock .......... $...
-
Assume that after initial recognition, Blossom uses the revaluation model (asset adjustment method) to measure its intangible assets. Prepare the entries required on December 31, 2025, December 31,...
-
Manager's Hot Seat Negotiation: Thawing the Salary Freeze from the video create a report. What could Katherine have done or said differently?
-
A company has three team leaders available for assignment thrRRAHAtover The estimated task completion times in hours are as to follows: Customers Team Leades 123 20 25 19 19 28 15 16 24 13 A B C...
-
Suppose that debit cards, ATMs, ACH transactions, and other forms of electronic funds transfers did not exist. How would this change the way you shop and pay bills? How would transactions costs in...
-
What is the appeal of the eurocurrency market?
-
What is the appeal of trade shows and trade missions for small and medium-sized companies?
-
Governments influence the international flow of trade and foreign direct investment in many ways, such as by forming regional economic groups. Select two market entry modes and answer the question:...
-
What is it called when a company grants another the right to use intangible property for a limited period of time?
-
7. The formation constant (K) for Cu(NH 3) 4^2+ is 5 * 10 ^ 13 A) Write the balanced reaction. Cu ^2 + (a 9 ) + 4NH 3 (a 9 ) Cu(NH 3 ) 4 (aq); Kf = [Cv(NH 3) 4 [Cv^ 24 ] Cnv b) Using the reaction and...
-
Refer to the data for problem 13-36 regarding Long Beach Pharmaceutical Company. Required: Compute each division's residual income for the year under each of the following assumptions about the...
-
1. How has an understanding of consumer behavior helped Groupon grow from 400 subscribers in Chicago in 2008 to 60 million subscribers in 40 countries today? 2. What is the Groupon Promise? How does...
-
What methods would you suggest to assess the potential commercial success for the following new products? (a) A new, improved ketchup; (b) A three-dimensional television system that took the company...
-
(a) Why might a marketing researcher prefer to use secondary data rather than primary data in a study? (b) Why might the reverse be true?
-
1. Describe the bases of power held by Dr. Jamie Thompson. Describe the bases of power held by Dr. Elizabeth Clarke. 2. What activities and people have contributed to Jaime Thompsons power? What...
-
What is the current in the wire in Figure Q22.1? 1.0 VR + 1.0-1.0V + FIGURE Q22.1
-
Electroplating uses electrolysis to coat one metal with another. In a copper-plating bath, copper ions with a charge of +2e move through the electrolyte from the copper anode to the cathode the metal...

Study smarter with the SolutionInn App


