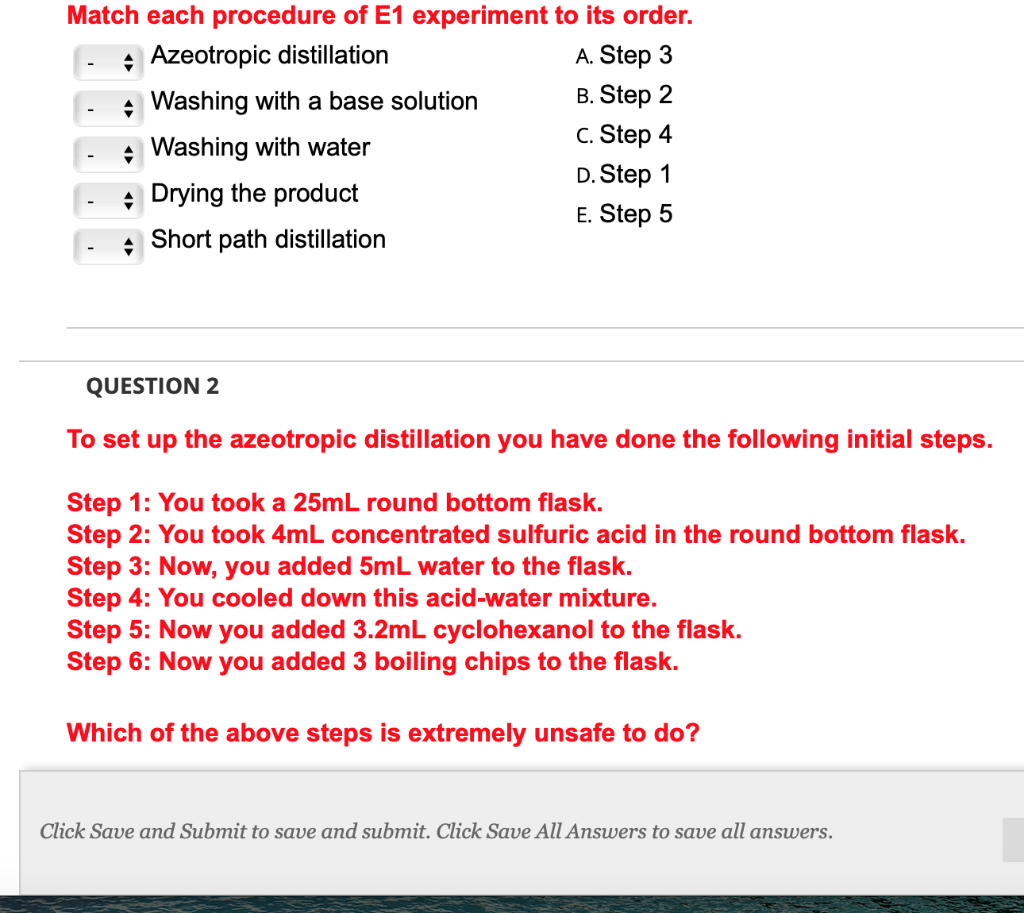
Match each procedure of E1 experiment to its order. Azeotropic distillation A. Step 3 Washing with...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Match each procedure of E1 experiment to its order. Azeotropic distillation A. Step 3 Washing with a base solution B. Step 2 Washing with water c. Step 4 D. Step 1 Drying the product E. Step 5 Short path distillation QUESTION 2 To set up the azeotropic distillation you have done the following initial steps. Step 1: You took a 25mL round bottom flask. Step 2: You took 4mL concentrated sulfuric acid in the round bottom flask. Step 3: Now, you added 5mL water to the flask. Step 4: You cooled down this acid-water mixture. Step 5: Now you added 3.2mL cyclohexanol to the flask. Step 6: Now you added 3 boiling chips to the flask. Which of the above steps is extremely unsafe to do? Click Save and Submit to save and submit. Click Save All Answers to save all answers. Match each procedure of E1 experiment to its order. Azeotropic distillation A. Step 3 Washing with a base solution B. Step 2 Washing with water c. Step 4 D. Step 1 Drying the product E. Step 5 Short path distillation QUESTION 2 To set up the azeotropic distillation you have done the following initial steps. Step 1: You took a 25mL round bottom flask. Step 2: You took 4mL concentrated sulfuric acid in the round bottom flask. Step 3: Now, you added 5mL water to the flask. Step 4: You cooled down this acid-water mixture. Step 5: Now you added 3.2mL cyclohexanol to the flask. Step 6: Now you added 3 boiling chips to the flask. Which of the above steps is extremely unsafe to do? Click Save and Submit to save and submit. Click Save All Answers to save all answers.
Expert Answer:
Answer rating: 100% (QA)
Azeotropic distillation C Step 4 Washing with a base solution B Step 2 Was... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
Concentrated sulfuric acid reacts with sodium iodide to produce molecular iodine, hydrogen sulfide, and sodium hydrogen sulfate. Write a balanced equation for the reaction.
-
A weak acid is added to a concentrated solution of hydrochloric acid. Does the solution become more or less acidic?
-
The concentration of commercially available concentrated sulfuric acid is 98.0 percent by mass, or 18 M. Calculate the density and the molality of the solution.
-
A fast-food restaurant operates both a drive through facility and a walk-in facility. On a randomly selected day, let X and Y, respectively, be the proportions of the time that the drive-through and...
-
The Nippon Company purchases 80,000 pumps annually from Xing Brothers, Inc. The price has increased each year and reached $68 per unit last year. Because the purchase price has increased...
-
Patterson Company is considering two competing investments. The first is for a standard piece of production equipment. The second is for computer- aided manufacturing (CAM) equipment. The investment...
-
Florence Kundrat owns Discount Fashions. She is contemplating the purchase of a soda machine, which would be used to sell soft drinks to customers for $0.75 each. The following estimates are...
-
Donna Shader, manager of the Winter Park Hotel, is considering how to restructure the front desk to reach an optimum level of staff efficiency and guest service. At present, the hotel has five clerks...
-
PLEASE ANSWER AS SOON AS POSSIBLE. A fund had an opening value of ?2m on 1 January 2005 and closing value of ?2.4m on 1 January 2008. A cashflow of ?500k was received on 1 July 2005 and ?500k ...
-
Balance sheets for P Company and S Company on August 1, 2019, are as follows: Required: Prepare a workpaper for a consolidated balance sheet for P Company and its subsidiary on August 1, 2019, taking...
-
The supplier selection process is a critical activity within the operations of a company. Once the outsourcing decision is taken, a process aimed at analyzing and choosing the appropriate suppliers...
-
Rearrage the equation to isolate a . ( a + b / c ) ( d - e ) = f
-
Actual and estimated data for manufacturing overhead, direct labor cost, direct labor hours, and machine hours for the most recent fiscal year are summarized here: Direct labor cost Manufacturing...
-
which point is nearest the x-axis P. (-3,4) B. (5,-2) L. (4,5) D. 1-5,3
-
Olympus Gear, a company specializing in outdoor equipment, faces a challenge. Their division, "Guardian Defense," manufactures protective armor for security personnel. Xena, the division manager,...
-
You have a restaurant with 45 tables divided into five sections of equal size. You are only open starting at 17:00 Tuesday to Sunday, closing each day at 22:00, with last seating at 21:00. Your track...
-
Exercise 12-7A (Algo) The direct versus the indirect method of determining cash flows from operating activities LO 12-1, 12-2 The following accounts and corresponding balances were drawn from Jogger...
-
Suppose you won a financial literacy competition and are given FJS10000 to invest, with the condition that investment can be done either in, i) Invest in Unit trust of Fiji or Invest in Fijian...
-
In 2012, Margaret and John Murphy are married taxpayers who file a joint tax return with AGl of $25,000. During the year they incurred the following expenses: Hospitalization insurance...
-
Diane is a single taxpayer who qualifies for the earned income credit. Diane has two qualifying children who are 3 and 5 years old. During 2012, Diane's wages are $17,100 and she receives dividend...
-
Olive Corporation was formed and began operations on January 1, 2012. The corporation's income statement for the year and the balance sheet at year-end are presented below. The corporation made...
-
Demonstrate the density property of rational numbers by finding a rational number between \(\frac{27}{13}\) and \(\frac{21}{10}\).
-
Correctly apply the rules for the order of operations to accurately compute \(\left(\frac{3}{16}+\frac{7}{16} ight)^{2}+\frac{1}{5} \div \frac{3}{10}\).
-
Danny, a nutritionist, is designing a diet for her client, Callum. Danny determines that Callum's diet should be \(30 \%\) protein. If Callum consumes 2,400 calories per day, how many calories of...

Study smarter with the SolutionInn App


