1. Calculate the concentration, in grams of solute per 100 g of water, of a solution...
Fantastic news! We've Found the answer you've been seeking!
Question:
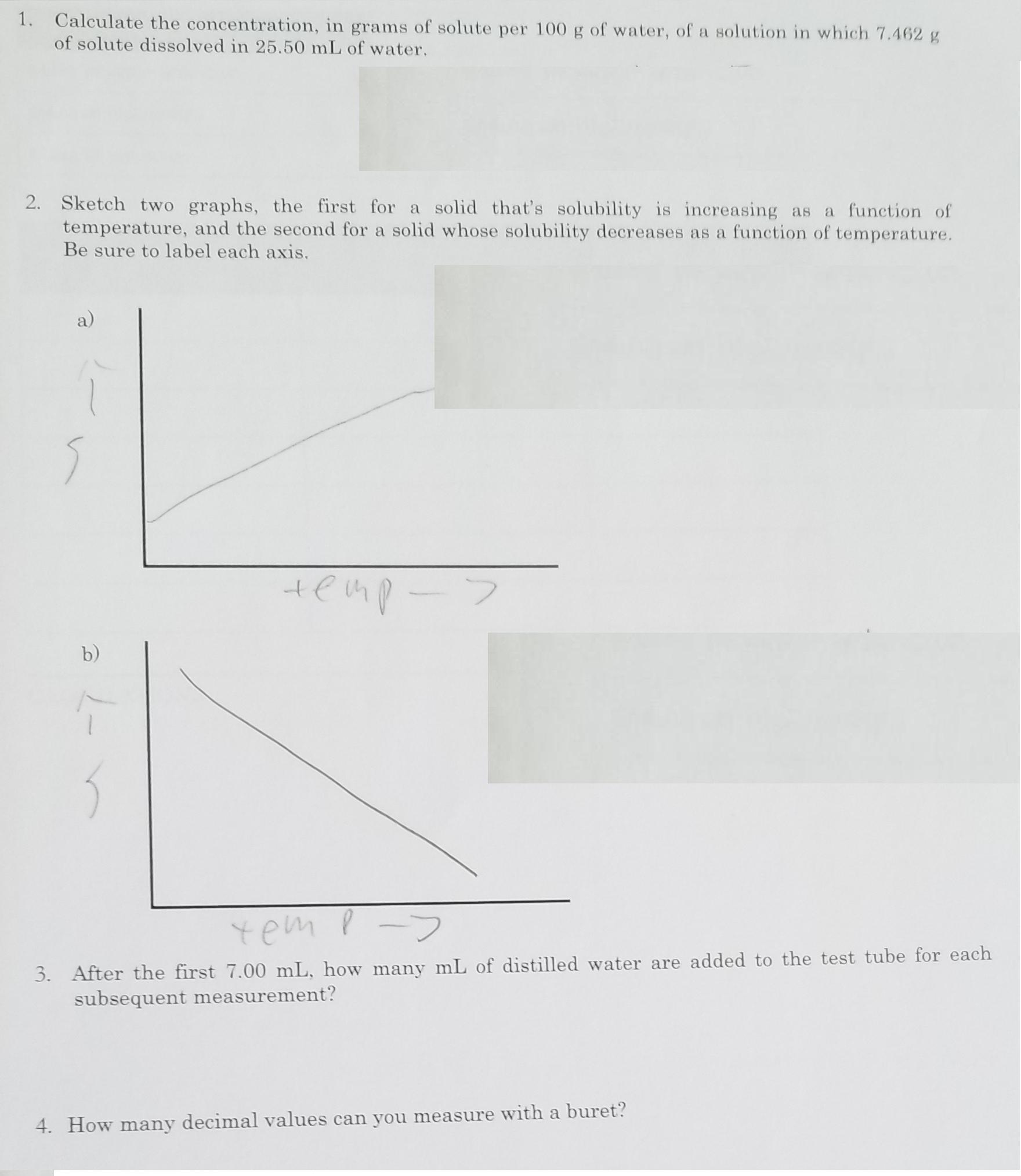
Transcribed Image Text:
1. Calculate the concentration, in grams of solute per 100 g of water, of a solution in which 7.462 g of solute dissolved in 25.50 mL of water. 2. Sketch two graphs, the first for a solid that's solubility is increasing as a function of temperature, and the second for a solid whose solubility decreases as a function of temperature. Be sure to label each axis. a) b) yem p-> 3. After the first 7.00 mL, how many mL of distilled water are added to the test tube for each subsequent measurement? 4. How many decimal values can you measure with a buret? 1. Calculate the concentration, in grams of solute per 100 g of water, of a solution in which 7.462 g of solute dissolved in 25.50 mL of water. 2. Sketch two graphs, the first for a solid that's solubility is increasing as a function of temperature, and the second for a solid whose solubility decreases as a function of temperature. Be sure to label each axis. a) b) yem p-> 3. After the first 7.00 mL, how many mL of distilled water are added to the test tube for each subsequent measurement? 4. How many decimal values can you measure with a buret?
Expert Answer:
Answer rating: 100% (QA)
B 1 Given Mass of solute 7462 g Volume of solution 2550 ml To find Concentration in grams of solute ... View the full answer

Related Book For 

Fundamentals of Analytical Chemistry
ISBN: 978-0495558286
9th edition
Authors: Douglas A. Skoog,Donald M. West, F. James Holler, Stanley R. Crouch
Posted Date:
Students also viewed these chemistry questions
-
Graph each function. Be sure to label any intercepts. f(x) = V16 + 4x
-
Graph each function. Be sure to label any intercepts. f(x) = -V9 + 9x
-
Graph each function. Be sure to label any intercepts. f(x) = -V-25 + x
-
With the CARES Act costing over $2 trillion and the federal government facing a significant decline in tax revenue, explain the effectiveness of fiscal policy using the three claims outlined in the...
-
On November 1, 2009, Davidson Co. received a $5,000, 10%, 4-month note from Chris Spent in exchange for his open account receivable. Davidsons scal year ends on December 31. Required: (a) Prepare the...
-
When applying absolute value models, constant growth models tend to be used when. a. There is no need to forecast future cash flows. b. Required returns are constant. c. Expected cash flows are...
-
Gamma Oil Company sold or used the gas produced on Lease A during January as follows: a. 300 Mcf used as fuel to operate lease equipment b. 800 Mcf sold to R Company at $12/Mcf Assume a 1/7 RI and a...
-
As the auditor of Clearwater County you learn that various assets are subject to spending constraints. Indicate how each of the following constraints would affect the countys reported fund balance...
-
For Questions 19 and 20, refer to the figures. 19. Given: AC = DF, AB DE, m mLD Which can be concluded by the Hinge Theorem? A AABC = ADEF B BC= EF A B D D BC > EF E CBC
-
A solar collector design consists of several parallel tubes each enclosed concentrically in an outer tube which is transparent to solar radiation. The tubes are thin walled with diameter of the inner...
-
Calculate discount factors from project A . Project A 10% discount Present value Year Cash flow () factor = cash flow () 0 -1500 1 = -1 500 1 +500 0.9091 = + 454.55 2 +800 0.8264 + 661.12 3 +1 000...
-
FastTrack Motors assembles and sells motor vehicles and uses standard costing. Actual data and variable costing and absorption costing income statements relating to April and May 2014 2014 are as...
-
An investment promises the following cash flow stream: -$750 now Year 0; $2,450 at the end of Year 1; $3,175 at the end of Year 2; $0 at the end of year 3;and $4,400 at the end of Year 4. The project...
-
The competition between Bombardier and Embraer had the far-reaching effects, even affecting relations between Canada and Brazil. Comment on the issues in the Bombardier - Embraer face-off, and...
-
Evaluate the idea that a world hegemon creates stability in the global system. What are the pros and cons of this theory? If , as some political scientists predict, U . S . hegemony is coming to an...
-
Marigold, Inc. is facing a problem with their 4th quarter absorption costing net operating income on December 25. Their net operating income target is $350,000 and the data so far is as follows: ...
-
2. Preparing Formal Business Reports A formal report may be defined as a document in which a writer Formal reports are used to assist in the What is the first step in formal report writing? Determine...
-
Software Solution is family-owned business that has been in operation for more than 15 year. The board of directors is comprised of mainly family members, plus a few professionals such as an...
-
What mass of AgI can be produced from a 0.512-g sample that assays 20.1% AlI3?
-
A 1.509-g sample of a Pb/Cd alloy was dissolved in acid and diluted to exactly 250.0 mL in a volumetric flask. A 50.00-mL aliquot of the diluted solution was brought to a pH of 10.0 with a NH4+/NH3...
-
In contrast to many salts, calcium sulfate is only partially dissociated in aqueous solution: The solubility-product constant for CaSO4 is 2.6 10-5. Calculate the solubility of CaSO4 in (a) water...
-
How does activity-based management (ABB) build on ABC?
-
Explain the difference between a traditional hotel accounting approach and that used in CPA.
-
How can CPA aid management decision making?

Study smarter with the SolutionInn App


