Part A The equilibrium constant, K, of a reaction at a particular temperature is determined by...
Fantastic news! We've Found the answer you've been seeking!
Question:
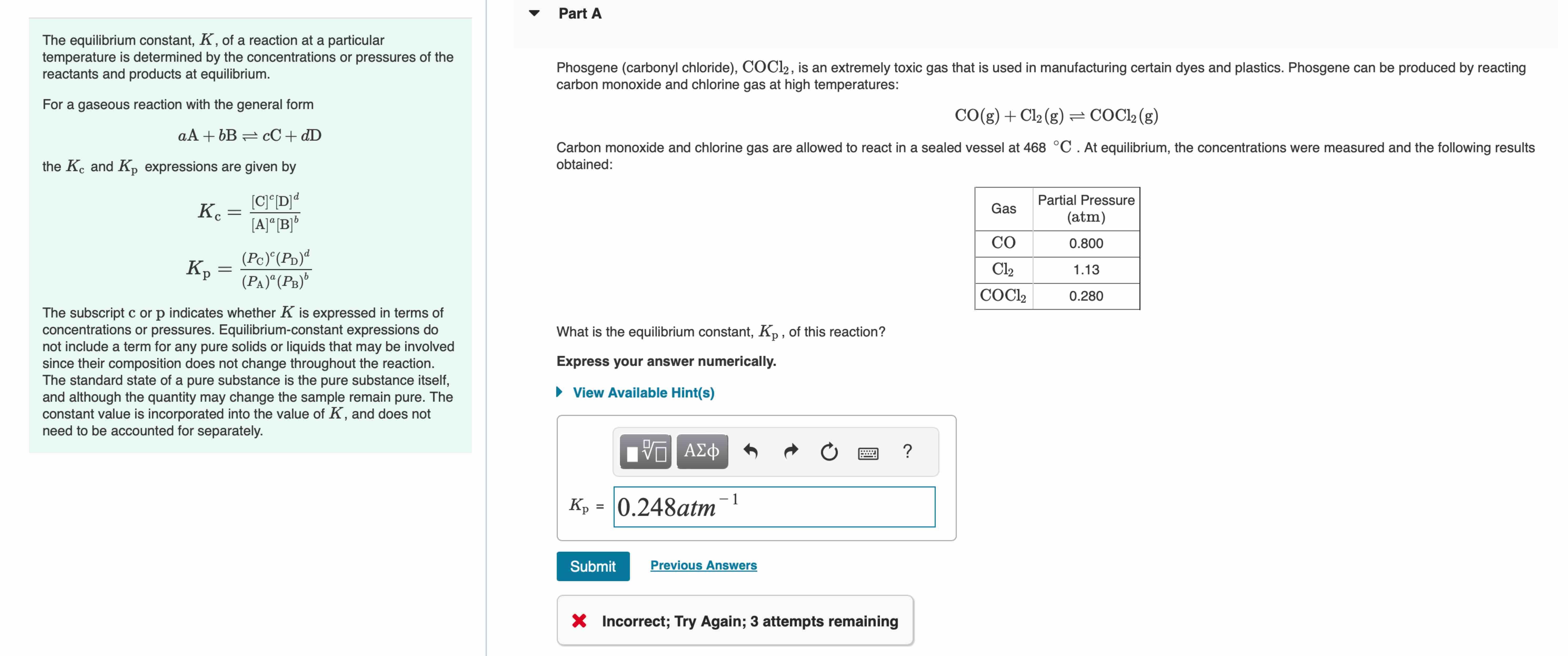
Transcribed Image Text:
Part A The equilibrium constant, K, of a reaction at a particular temperature is determined by the concentrations or pressures of the reactants and products at equilibrium. For a gaseous reaction with the general form aA+bB = cC+dD the Kc and Kp expressions are given by Kc K = [C]c[D] d [A][B]b (Pc) (PD)d (PA)a (PB) The subscript c or p indicates whether K is expressed in terms of concentrations or pressures. Equilibrium-constant expressions do not include a term for any pure solids or liquids that may be involved since their composition does not change throughout the reaction. The standard state of a pure substance is the pure substance itself, and although the quantity may change the sample remain pure. The constant value is incorporated into the value of K, and does not need to be accounted for separately. Phosgene (carbonyl chloride), COC12, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. Phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: CO(g) + Cl2(g) = COCl2(g) Carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 468 C. At equilibrium, the concentrations were measured and the following results obtained: What is the equilibrium constant, Kp, of this reaction? Express your answer numerically. View Available Hint(s) 0 ? Gas Partial Pressure (atm) CO 0.800 Cl 1.13 COCl2 0.280 1 Kp = 0.248atm Submit Previous Answers Incorrect; Try Again; 3 attempts remaining Part A The equilibrium constant, K, of a reaction at a particular temperature is determined by the concentrations or pressures of the reactants and products at equilibrium. For a gaseous reaction with the general form aA+bB = cC+dD the Kc and Kp expressions are given by Kc K = [C]c[D] d [A][B]b (Pc) (PD)d (PA)a (PB) The subscript c or p indicates whether K is expressed in terms of concentrations or pressures. Equilibrium-constant expressions do not include a term for any pure solids or liquids that may be involved since their composition does not change throughout the reaction. The standard state of a pure substance is the pure substance itself, and although the quantity may change the sample remain pure. The constant value is incorporated into the value of K, and does not need to be accounted for separately. Phosgene (carbonyl chloride), COC12, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. Phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: CO(g) + Cl2(g) = COCl2(g) Carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 468 C. At equilibrium, the concentrations were measured and the following results obtained: What is the equilibrium constant, Kp, of this reaction? Express your answer numerically. View Available Hint(s) 0 ? Gas Partial Pressure (atm) CO 0.800 Cl 1.13 COCl2 0.280 1 Kp = 0.248atm Submit Previous Answers Incorrect; Try Again; 3 attempts remaining
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
1.5 inches The 0.35 lbf ball is shot from the spring device shown. The spring has a stiffness k = 12 lbf/in. The plate reduces the spring to 1.5 inches in length, when its neutral length is 4 inches....
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
On February 1, 2020, Sheridan Company sells merchandise on account to Carla Vista Company for $6490. The entry to record this transaction by Sheridan Company is Sales Revenue Accounts Payable Notes...
-
Obtain the complex power delivered by the source in the circuit of Fig. 11.75. 2/30 2
-
How do memory management techniques such as virtual memory, paging, and segmentation contribute to the optimization of memory utilization and address space management in complex operating...
-
Consider the following financial statements for Benjamin Company. During 2019, management obtained additional bond financing to enlarge its production facilities. The company faced higher production...
-
Rhiener Corporation initiated a performance-based employee stock option plan on January 1, 2010. The performance base for the plan is net sales in the year 2012. The plan provides for stock options...
-
Assess the government and private plans to overcome financial, technical, and administrative barriers to implementing regional health information organizations and national health information...
-
Some believe that International Financial Reporting Standards (IFRS) provide too many choices within its accounting guidance. Is this a possible concern in the area of cash flow reporting in your...
-
Imagine a scenario where a major financial institution, like the Federal Reserve, adopts a more aggressive approach to regulating financial markets to prevent another economic crisis. How would these...
-
The Project Management Book of Knowledge PMBoK Guide describes a PMIS as "An information system consisting of the tools and techniques used to gather, integrate, and disseminate the outputs of...
-
Check my work Equity Dividends Revenue a. The company completed consulting work for a client and immediately collected $5,500 cash. b. The company completed commission work for a client and sent a...
-
Answer the questions based on the scenario: It is a hot and sunny day on the 15th of July 2016, and Windward Air flight #1234 from Beijing has just landed at Edmonton's International Airport. When...
-
Refer to the following lease amortization schedule. The 1 0 payments are made annually starting with the beginning of the lease. Title does not transfer to the lessee and there is no purchase option...
-
MXP Co has set a minimum cash account balance of $75,000. The average cost to the company of making deposits or selling securities is $50 per transaction and the standard deviation of its cash flows...
-
What is removed during each of the three stages of wastewater treatment: primary, secondary, and tertiary? During which state would you expect items to be recovered that were accidentally flushed,...
-
Which of the following techniques involves the creation and use of a pair of public and private keys? a. hashing c. symmetric encryption b. asymmetric encryption d. key escrow
-
Which of the following is a preventive control? a. penetration testing c. training b. patch management d. log analysis
-
The approach to perimeter defense that involves examining only information in the packet header of each individual IP packet is referred to as a. deep packet inspection c. stateful packet filtering...

Study smarter with the SolutionInn App


