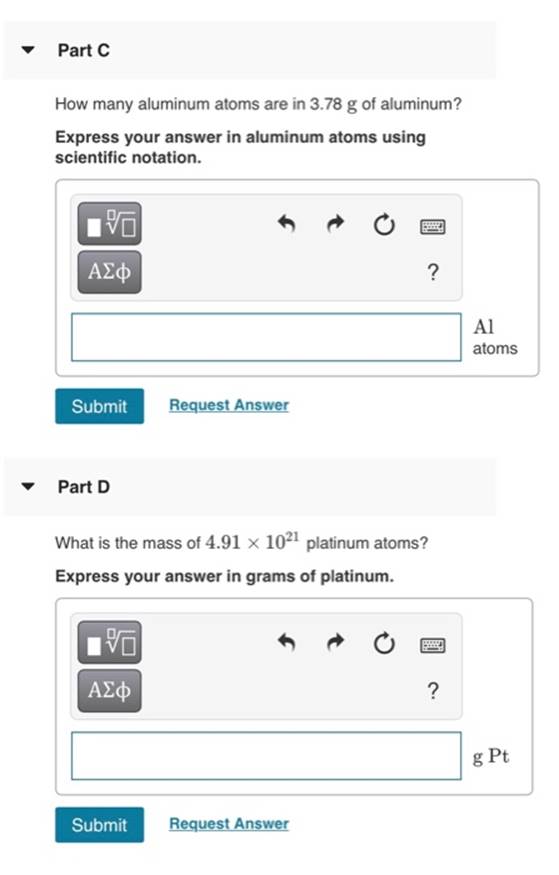
Part C How many aluminum atoms are in 3.78 g of aluminum? Express your answer in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Part C How many aluminum atoms are in 3.78 g of aluminum? Express your answer in aluminum atoms using scientific notation. ΑΣφ ? Al atoms Submit Request Answer Part D What is the mass of 4.91 x 1021 platinum atoms? Express your answer in grams of platinum. ΑΣφ ? g Pt Submit Request Answer Part C How many aluminum atoms are in 3.78 g of aluminum? Express your answer in aluminum atoms using scientific notation. ΑΣφ ? Al atoms Submit Request Answer Part D What is the mass of 4.91 x 1021 platinum atoms? Express your answer in grams of platinum. ΑΣφ ? g Pt Submit Request Answer
Expert Answer:
Answer rating: 100% (QA)
Answer Past C As wrdas mass of Al 26981 u Griven mase of Af 3789 Molee f Al 348g O 14 moles 26... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
What is the mass of a 1.00-Ci 14/8C source?
-
What is the mass of the solid NH4Cl formed when 73.0 g of NH3 are mixed with an equal mass of HCl? What is the volume of the gas remaining, measured at 14.0C and 752 mmHg? What gas is it?
-
What is the mass density of air at P = 1.0 atm and T = (a) 10 C and (b) 30 C? The average molecular mass of air is approximately 29 u.
-
Common structural steel can be represented on drawings using a letter, immediately followed by a number, then another number (for example, S14x30). The two numbers for a steel member W16x60 designate...
-
A corporation reacquires 60,000 shares of its own $10 par common stock for $3,000,000, recording it at cost. a. What effect does this transaction have on revenue or expense of the period? b. What...
-
What are the oxidation numbers of the nitrogen atoms in these substances? a. N2 b. NH3 c. NO d. N2O e. NO2 f. N2O4 g. N2O5 h. NaNO3
-
It has been argued that to be classified as a professional one must offer services to the public rather than simply be an employee of an organization. As such, most employees of an organization,...
-
Refer to the data for the Ferre Publishing Company in Exercise 4B3. In Exercise 4B3, The Ferre Publishing Company has three service departments and two operating departments. Selected data from a...
-
At what sample size do we begin minimizing the error in the standard error of the mean? 10 30 O 50 100
-
? ?? Journalize the closing entries. Include Posting References, using the account numbers in your spreadsheet. Refer to your answers from Chapter 4, Part 1 to complete this part. If an amount box...
-
When water is produced in a combustion process, the enthalpy of combustion depends on whether water ends up as a liquid or a vapor. The difference may be significant because the latent heat of...
-
The downward distance d that an object falls in a time interval t if starting at rest is dat. On the Moon, a rock falls 10.0 m in 3.50 s. What time interval t is needed for it to fall 15.0 m,...
-
Kiwidale Dairy is considering purchasing a new ice-cream maker. Two models, Smoothie and Creamy, are available and their information is given below. Answer parts (a) and (b) below. First cost Service...
-
You decide to open a pizza parlor. The ingredients require that you charge $4.50 for a 7.0-in.-diameter pizza. How large should you make a pizza whose price is $10.00, assuming the cost is based...
-
Organize an easter event, Reflect and describe an example of when you sought to share and seek information with colleagues or a supervisor. How did this help to maximize cooperation? How did this...
-
Bernard co. has 9% coupon bonds on the market that have 13 years left to maturity. The bonds will make annual payments. If the YTM on these bonds is 6%, what is the current bond price (in $ dollars)?...
-
[SECTION 1 TOTAL - 50 MARKS] SECTION 2 QUESTION 1 Read the following Case-Study titled "Troublesome Technology" carefully and answer the questions that follow: Troublesome technology Failure to...
-
In July 2013, cnet.com listed the battery life (in hours) and luminous intensity (i. e., screen brightness, in cd/m2) for a sample of tablet computers. We want to know if screen brightness is...
-
Making mayonnaise involves beating oil into small droplets in water, in the presence of egg yolk. What is the purpose of the egg yolk?
-
Acetic acid is a polar molecule and can form hydrogen bonds with water molecules. Therefore, it has a high solubility in water. Yet acetic acid is also soluble in benzene (C6H6), a nonpolar solvent...
-
Acetic acid is a weak acid that ionizes in solution as follows: If the freezing point of a 0.106 m CH 3 COOH solution is 20.203C, calculate the percent of the acid that has undergone ionization....
-
Define flowcharting.
-
Flowchart symbols represent what aspect of a system?
-
List and draw the basic flowchart symbols.

Study smarter with the SolutionInn App


