The most abundant radon isotope is radon-222, which is an alpha emitter with a half-life of...
Fantastic news! We've Found the answer you've been seeking!
Question:

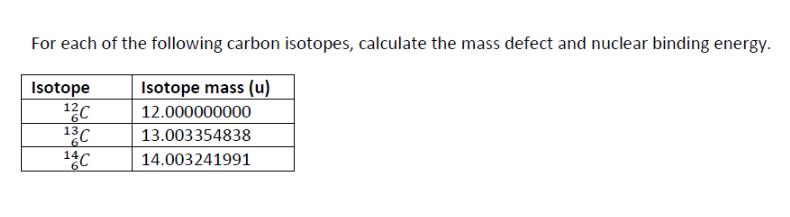
Transcribed Image Text:
The most abundant radon isotope is radon-222, which is an alpha emitter with a half-life of 3.8 days. The air in a house basement in Canada is found to have a radon level of 500 Bq m³. The World Health Organization's safe limit for radon is 100 Bq m³. How long will it take for the radon level to decay to 100 Bq m³ in a sample of air from this basement? An archaeologist uncovers a wooden object at an archaeological site. The object is found to have a carbon-14 decay rate of 0.11 disintegrations per second per gram of carbon. How old is the object? (Living organisms have a decay rate of 0.255 s¹ g¹, and t₁/2 = 5730 y for C-14.) Thorium-232 has a radioactive decay series that ultimately produces lead-208. What is the expected ratio of 202Pb/232Th in a meteorite that is approximately 3.0 × 109 years old? (The half-life of thorium-232 is 1.4 x 1010 y.) For each of the following carbon isotopes, calculate the mass defect and nuclear binding energy. Isotope 12C Isotope mass (u) 12.000000000 13C 13.003354838 6 14C 14.003241991 The most abundant radon isotope is radon-222, which is an alpha emitter with a half-life of 3.8 days. The air in a house basement in Canada is found to have a radon level of 500 Bq m³. The World Health Organization's safe limit for radon is 100 Bq m³. How long will it take for the radon level to decay to 100 Bq m³ in a sample of air from this basement? An archaeologist uncovers a wooden object at an archaeological site. The object is found to have a carbon-14 decay rate of 0.11 disintegrations per second per gram of carbon. How old is the object? (Living organisms have a decay rate of 0.255 s¹ g¹, and t₁/2 = 5730 y for C-14.) Thorium-232 has a radioactive decay series that ultimately produces lead-208. What is the expected ratio of 202Pb/232Th in a meteorite that is approximately 3.0 × 109 years old? (The half-life of thorium-232 is 1.4 x 1010 y.) For each of the following carbon isotopes, calculate the mass defect and nuclear binding energy. Isotope 12C Isotope mass (u) 12.000000000 13C 13.003354838 6 14C 14.003241991
Expert Answer:
Answer rating: 100% (QA)
1 Radon222 decay in the basement Radon222 has a halflife of 38 days The initial radon level is 500 B... View the full answer

Related Book For 

Auditing a risk based approach to conducting a quality audit
ISBN: 978-1133939153
9th edition
Authors: Karla Johnstone, Audrey Gramling, Larry Rittenberg
Posted Date:
Students also viewed these finance questions
-
Find the distance between the two points (-2,5) and (3,7). 5 5 29 3
-
What is f'(x) if f(x) = x64? 64x63 64x +63 63x
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Do firms follow the same steps for impairment testing of finite- and indefinite- life intangible assets ? Explain.
-
Aida created the following double bar graph. It illustrates the actual amount spent this year for household expenses per month and the budgeted amounts for the following year. The following year's...
-
Catherine Ruby, a doctoral student at New York University, conducted an online survey to ascertain the reasons that international students chose to attend graduate school in the United States. One of...
-
Public corporations are required not only to communicate with their stockholders by means of an annual report but also to submit an annual report to the Securities and Exchange Commission (SEC). The...
-
Doris Stewart started her practice as a design consultant on September 1, 2012. During the first month of operations, the business completed the following transactions: Sep 1 Received $42,000 cash...
-
Can you please help? I have attached the instructions on the word doc and the spreadsheet. Please let me know if you have any questions. Introduction: You are the senior financial analyst for Fosbeck...
-
A company manufactures a single product. Budget and standard cost details for next year include: Selling price per unit ................................$24.00 Variable production cost per unit...
-
1 The following entities issue bonds to engage in long-term borrowing except a. the federal government. b. state and local governments. c. corporations. d. individuals 2 If a bond pays interest...
-
Setup and evaluate the integral (using exact values) for the area of the region bounded by the graphs of the functions y = x and y = -x-x+18. 8 Area = Evaluate the integral. 1 15 Enter an exact...
-
You get a 30-year, $280,000 mortgage at 6.4% interest.Calculate the amount of the final payment on this mortgage.
-
Imagine that you are Leila Velez, the CEO of Beleza Natural, and you have to decide how to grow the company to reach your goal. What would you do ? If you decided to make Beleza\'s products available...
-
Makina Company manufactures engines on a cost-plus basis. The cost of a particular machine follows: Direct materials, P400,000; Direct Labor, P300,000; Supervisor's salary, P40,000; Fringe benefits...
-
A firm sells its products on terms of net 30 at $205 per unit. The present value of production cost is $135 per unit. The opportunity cost is 1% per month, and there are 30 days per month. It is...
-
10. value 5.00 points July 1 Purchased merchandise from Black Company for $6,900 under credit terms of 2/15, n/30, FOB shipping point, invoice dated July 1. 2 Sold merchandise to Coke Co. for $950...
-
Per Bag Direct materials: 25 pounds of CWhiz-2000 @ $0.08/lb. = $ 2.00 Direct labor: 0.05 hour @ $32.00/hr. = $ 1.60 The company manufactured 100,000 bags of Cheese-Be-Good in December and used...
-
Match the following assertions with their associated description: (a) Existence or occurrence, (b) Completeness, (c) Rights and obligations, (d) Valuation or allocation, (e) Presentation and...
-
On February 28, 2014, Stu & Dent, LLP completed the audit of Shylo Ranch, Inc. (a public company) for the year ended December 31, 2013. A recent fire destroyed the accounting records concerning the...
-
Why is it important to assess whether potential misclassifications in the statement of cash flows are material?
-
Use your graph to find a route that the principal can take beginning at the Front Office in room \(A\), visiting each room exactly once (without passing by another room), and ending at room \(F\),...
-
Mellon Bank is a large bank holding company. Selected accounts from the companys 2004 annual report are as follows (in millions):9 Cash and Due from Banks $2,775 Securities Available for Sale $13,376...
-
You have been promoted recently and now have access to the firms monthly financial statements. Business is good. Revenues are increasing rapidly, and income is at an all-time high. The balance sheet...

Study smarter with the SolutionInn App


