Apply the Gibbs phase rule to determine the number of independent primary reactions needed to calculate...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
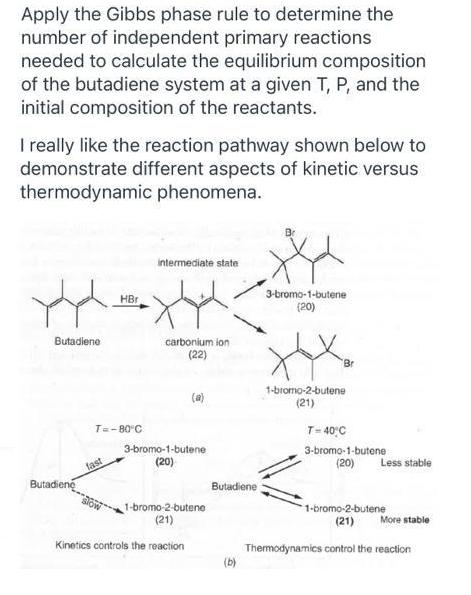
Apply the Gibbs phase rule to determine the number of independent primary reactions needed to calculate the equilibrium composition of the butadiene system at a given T, P, and the initial composition of the reactants. I really like the reaction pathway shown below to demonstrate different aspects of kinetic versus thermodynamic phenomena. intermediate state 3-bromo-1-butene (20) HBr Butadiene carbonium ion (22) Br 1-bromo-2-butene (21) (a) T=-80 C T- 40°C 3-bromo-1-butene 3-bromo-1-butene (20) fast (20) Less stable Butadiene Butadiene 1-bromo-2-butene (21) 1-bromo-2-butene (21) More stable Kinetics controls the reaction Thermodynamics control the reaction (b) Apply the Gibbs phase rule to determine the number of independent primary reactions needed to calculate the equilibrium composition of the butadiene system at a given T, P, and the initial composition of the reactants. I really like the reaction pathway shown below to demonstrate different aspects of kinetic versus thermodynamic phenomena. intermediate state 3-bromo-1-butene (20) HBr Butadiene carbonium ion (22) Br 1-bromo-2-butene (21) (a) T=-80 C T- 40°C 3-bromo-1-butene 3-bromo-1-butene (20) fast (20) Less stable Butadiene Butadiene 1-bromo-2-butene (21) 1-bromo-2-butene (21) More stable Kinetics controls the reaction Thermodynamics control the reaction (b)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A thermodynamic system undergoes a cyclic process as shown in Fig. 19.24. The cycle consists of two closed loops: I and II. (a) Over one complete cycle, does the system do positive or negative work?...
-
A thermodynamic system is taken from state A to state B to state C., and then back to A, as shown in the p-V diagram of Figure a. The vertical scale is set by ps = 40Pa, and the horizontal scale is...
-
A system goes from state 1 to state 3 as shown on the TS diagram in Fig. 12.25. (a) The heat transfer for the process going from state 2 to state 3 is (1) positive, (2) zero, (3) negative. Explain....
-
After researching Best Buy common stock, Sally Jackson is convinced the stock is overpriced. She contacts her account executive and arranges to sell short 200 shares of Best Buy. At the time of the...
-
Suppose you are a manager in a manufacturing company. Your accountant has just presented you with a very detailed cost analysis for a decision about whether to outsource or make a component of a...
-
Consider the following transactions that occurred in February 2012 for Gems, Inc. Feb 3 Purchased inventory on terms 1/5, n/eom, $2,000. 4 Purchased inventory for cash of $1,600. 6 Returned $600 of...
-
Assuming they have the same (positive) expected rate of return, which of the following investments should be cheapest? a. One that promises to pay you $100 in five years. b. One that promises to pay...
-
Plastic Products, LLC, manufactures plastic beverage bottles. The division that manufactures water bottles for the North American market has two plants that operate 24 hours a day, 365 days a year....
-
As sales manager, John was given the following static budget report in the clothing department of Dunham Company for the month of October: DUNHAM COMPANY Clothing Department Static Budget Report...
-
sounds coming from the north side of her three-story house. Toward the end of the second day, she examined the exterior of her house and noticed two large holes located near the roof. To inspect the...
-
A stock has had returns of 17.92 percent, 12.44 percent, 6.56 percent, 28.30 percent, and -14.09 percent over the past five years, respectively. What was the holding period return for the stock? (Do...
-
Given the alternative approaches identified in this chapter, think of a possible hybrid approach (keeping in mind time, resource and people constraints).
-
Consider the authors classification of technology into the basic characteristics of data storage, transmission, and processing capacity, and properties of communication and decision aiding. Would you...
-
The author states that successful SISP seems to require users and line managers working in partnership with the IS function. Who should be involved in SISP and how should those involved be determined?
-
What is the IT productivity paradox? Does it exist in your view, and if so, to what extent is it sectorally based? Do you believe it will remain a problem in, say, 5 years?
-
This chapter raises the important issue of improving customer support. What lessons do you take from this when considering information systems strategy and planning?
-
Z plc has found that it can estimate future sales using time series analysis and regression techniques. The following trend equation has been derived: y 25,000+ 6,500x where: y is the total sales...
-
Extend Algorithms 3.4 and 3.5 to include as output the first and second derivatives of the spline at the nodes.
-
Three 25.0-L flasks are placed next to each other on a shelf in a chemistry stockroom. The first flask contains He at a pressure of 1.0 atm, the second contains Xe at 1.50 atm, and the third contains...
-
Iron(II) ion is oxidized by hydrogen peroxide in acidic
-
Arrange the following substances in order of increasing melting point. Si H-C-O-H H-C-C-H NaCl
-
What is the cost of net debt of a company that has no more shareholders equity equal to? And the cost of capital?
-
Is the cost of capital an accounting or financial concept?
-
What are we forgetting when we say that by increasing return on equity, the leverage effect of debt cannot increase value?

Study smarter with the SolutionInn App


