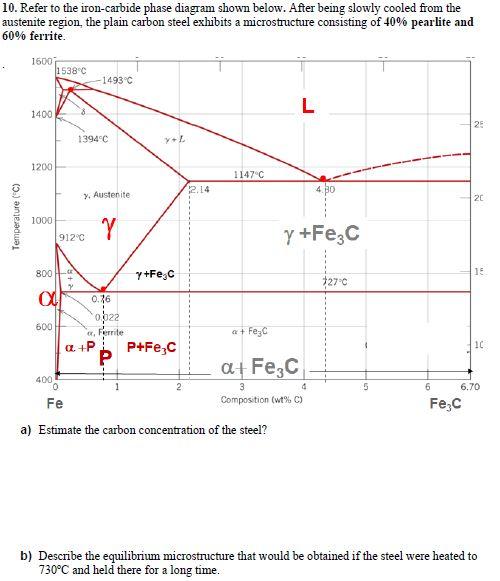
10. Refer to the iron-carbide phase diagram shown below. After being slowly cooled from the austenite...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
10. Refer to the iron-carbide phase diagram shown below. After being slowly cooled from the austenite region, the plain carbon steel exhibits a microstructure consisting of 40% pearlite and 60% ferrite. 1600 1538°C 1493°C L 1400 25 1394°C 1200 1147°C 2.14 y, Austenite 20 1000 912°C y +Fe,C 800 - y+Fe,C 15 127°C 0.76 op22 600 a, Ferrite a+ FeyC P+Fe,C 10 a+ Fe,C 400 3 6.70 Fe Composition (wt% C) Fe;C a) Estimate the carbon concentration of the steel? b) Describe the equilibrium microstructure that would be obtained if the steel were heated to 730°C and held there for a long time. Temperature ("C) 10. Refer to the iron-carbide phase diagram shown below. After being slowly cooled from the austenite region, the plain carbon steel exhibits a microstructure consisting of 40% pearlite and 60% ferrite. 1600 1538°C 1493°C L 1400 25 1394°C 1200 1147°C 2.14 y, Austenite 20 1000 912°C y +Fe,C 800 - y+Fe,C 15 127°C 0.76 op22 600 a, Ferrite a+ FeyC P+Fe,C 10 a+ Fe,C 400 3 6.70 Fe Composition (wt% C) Fe;C a) Estimate the carbon concentration of the steel? b) Describe the equilibrium microstructure that would be obtained if the steel were heated to 730°C and held there for a long time. Temperature ("C)
Expert Answer:

Related Book For 

Materials Science and Engineering An Introduction
ISBN: 978-0470419977
8th edition
Authors: William D. Callister Jr., David G. Rethwisch
Posted Date:
Students also viewed these accounting questions
-
The following information pertains to three portfolios over a 6-year period. Market returns (%) and the risk-free returns (%) are also provided. Period Market Return Risk free return Portfolio 1...
-
Below is a list of metals and alloys: Plain carbon steelMagnesium BrassZinc Gray cast ironTool steel PlatinumAluminum Stainless steelTungsten Titanium alloy Select from this list the one metal or...
-
The copper-silver phase diagram is shown in Figure 11-30. Copper has a higher melting point than silver. Figure 11-35 A phase diagram for elements A and B (for Problem 11-36). (a) Is copper element A...
-
Write a program that draws two circles with radius 15 pixels, centered at random locations, with a line connecting the two circles. The line should not cross inside the circles, as shown in Figure...
-
A certain compact disc player randomly plays each of 10 songs on a CD. Once a song is played, it is not repeated until all the songs on the CD have been played. In how many different ways can the CD...
-
State several financial ratios and their formulas that can be used by the auditor in applying analytical procedures to accounts receivable.
-
The most common choice for the appropriate estimation sampling period to use when estimating beta is to choose: a. 5 years of monthly data b. 5 years of yearly data c. 10 years of monthly data d. 10...
-
TL Division of Giant Bank has assets of $14.4 billion. During the past year, the division had profits of $1.8 billion. Giant Bank has a cost of capital of 8 percent. Ignore taxes. Required a. Compute...
-
Mountain View Plaza is a commercial office building with five tenants. These tenants and their monthly rental rate is listed below: Widget Corporation Dr. Spock First National Bank Comfy Office...
-
Using loop ACFDEA from Figure 14.10, and the data from Table 14.6, what is the (a) Misclosure in the X component? (b) Misclosure in the Y component? (c) Misclosure in the Z component? (d) Length of...
-
Beginning balances at 1 1 / 2 3 were as follows: Raw materials $ 7 0 , 0 0 0 Work in process $ 8 0 , 0 0 0 Finished goods $ 3 0 , 0 0 0 . Cash $ 1 5 , 0 0 0 The company applies overhead using a...
-
Consider a list A. We define an interleaved sublist to be a non-contiguous sublist taken from either only the even-indexed elements of L, or only the odd-indexed elements of L, in the order they...
-
Write a C program that will read two points (x1,y1) and (x2,y2). It will then calculate the slope and the y- intercept for the line that passes in those two points. m = x2-x1 c=y-m.x Where m is the...
-
1- Write a C++ program that prompts the user to enter five test scores (decimal) then: Prints the average of the scores. Converts the average to an integer value. (Use a constant to store the number...
-
The Star of India sapphire weighs 563 carats. If a carat equals 2.00 x 102 mg, what is the weight of the gemstone in pounds?
-
There is a temperature where the Fahrenheit scale and the Kelvin scale are numerically equal (hint: it is close to 500). Calculate this exact temperature using temperature conversions and the fact...
-
The numerator in the calculation of the ratio of fixed assets to long-term liabilities is Oa. Total Liabilities. Ob. Average Fixed Assets. Oc. Fixed Assets + Inventory. Od. Fixed Assets at Year-End.
-
A statistical study shows that the fraction of television sets of a certain brand that are still in service after x years is given by f (x) = e-0.15x. (a) What fraction of the sets are still in...
-
A brass alloy is known to have a yield strength of 275 MPa (40,000 psi), a tensile strength of 380 MPa (55,000 psi), and an elastic modulus of 103 GPa (15.0 106 psi). A cylindrical specimen of this...
-
The concentration of carbon in an iron-carbon alloy is 0.15 wt%. What is the concentration in kilograms of carbon per cubic meter of alloy?
-
Compare white and nodular cast irons with respect to (a) Composition and heat treatment, (b) Microstructure, and (c) Mechanical characteristics.
-
Why and how do tax authorities get involved in the transfer pricing situation?
-
Assume a VAT situation where the tax rate is 15 percent, with export sales exempt. The manufacturer does not purchase inputs on which VAT has been paid, and its net selling price to the wholesaler...
-
A Galex Com pain, a manufacturer of stereo systems, started its production in October 2002. For the preceding 3 years Galex had been a retailer of stereo systems. Alter a thorough survey ol stereo...

Study smarter with the SolutionInn App


