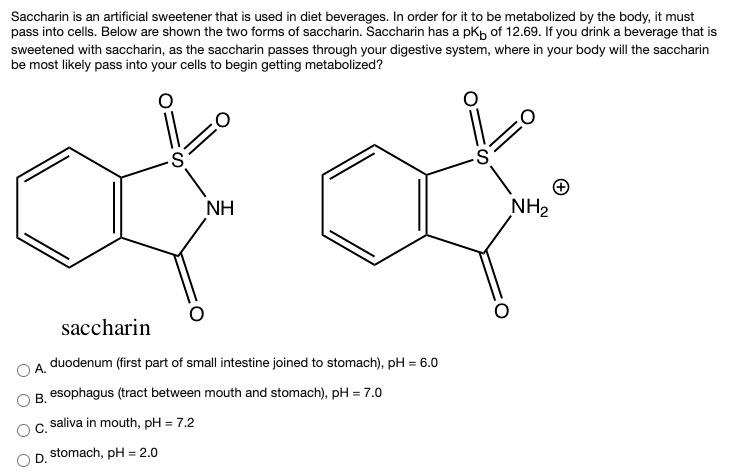
Saccharin is an artificial sweetener that is used in diet beverages. In order for it to...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Saccharin is an artificial sweetener that is used in diet beverages. In order for it to be metabolized by the body, it must pass into cells. Below are shown the two forms of saccharin. Saccharin has a pkb of 12.69. If you drink a beverage that is sweetened with saccharin, as the saccharin passes through your digestive system, where in your body will the saccharin be most likely pass into your cells to begin getting metabolized? NH NH2 saccharin duodenum (first part of small intestine joined to stomach), pH = 6.0 A. B. esophagus (tract between mouth and stomach), pH = 7.0 saliva in mouth, pH = 7.2 %3D C. stomach, pH = 2.0 D. Saccharin is an artificial sweetener that is used in diet beverages. In order for it to be metabolized by the body, it must pass into cells. Below are shown the two forms of saccharin. Saccharin has a pkb of 12.69. If you drink a beverage that is sweetened with saccharin, as the saccharin passes through your digestive system, where in your body will the saccharin be most likely pass into your cells to begin getting metabolized? NH NH2 saccharin duodenum (first part of small intestine joined to stomach), pH = 6.0 A. B. esophagus (tract between mouth and stomach), pH = 7.0 saliva in mouth, pH = 7.2 %3D C. stomach, pH = 2.0 D.
Expert Answer:
Answer rating: 100% (QA)
D stomach acid pH 2 pKb of saccharin 1269 this indicat... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Aspartame is an artificial sweetener that is 160 times sweeter than sucrose (table sugar) when dissolved in water. It is marketed as NutraSweet. The molecular formula of aspartame is C 14 H 18 N 2 O...
-
Xylitol is a sweetener that is used in sugarless chewing gum. Starting with an appropriate monosaccharide, outline a possible synthesis of xylitol.a OH OH HO OH OH Xylitol
-
Aspartame (below) is an artificial sweetener used in diet soft drinks and is marketed under many trade names, including Equal TM and Nutrasweet TM . In the body, aspartame is hydrolyzed to produce...
-
1-Kristen prefaces her speech by talking about her inability to save or invest money. For that reason, she does her speech on an online money management course and focuses on the skills she learned...
-
Amy, Beth, and Meg each own 100 of the 300 outstanding shares of Theta Corporation stock. Amy wants to sell her shares, which have a $40,000 basis and a $100,000 FMV. Either Beth and/or Meg can...
-
Foster Company initially records prepaid and unearned items in income statement accounts. Given Foster Companys practices, what is the appropriate adjusting entry for each of the following at...
-
Why is it necessary to sometimes redact portions of a document that is produced to the opposing side?
-
The following question illustrates the APT. Imagine that there are only two pervasive macroeconomic factors. Investments X, Y, and Z have the following sensitivities to these two factors: We assume...
-
A firm will need to take out a$200,000 loan 60days from now for a180-day interval. It purchases a call with X=4.1%.The call expires in 60days and the underlying is a180-day corporate loan rate. What...
-
The received signal in a digital base band system is either +A or -A, equally likely, for T-second contiguous intervals. However, the timing is off at the receiver so that the integration starts...
-
A particle moves in a straight line with position function 2r + 45, where t is measured in seconds and s in metres. Find the velocity of the particle at time t = a. find the velocities after 1 s, 2...
-
Consider and discuss how Lewins general theory of change underpins the complacency and leading change models (Exhibits 18.6 and 18.7). Exhibit 18.6 Exhibit 18.7 1. Establishing a sense of urgency 2....
-
What are the implications of behavioral finance?
-
You are asked to design a packed column to recover acetone from air continuously by absorption with pure water at \(289 \mathrm{~K}\). The air contains \(3.0 \mathrm{~mol} \%\) acetone, and a \(97...
-
Perhaps you have watched the game show known as Jeopardy. Contestants must prepare a question that is answered by a given prompt. It is your turn to play Jeopardy and your category is tools of...
-
Fill in the blanks to make the following statements correct. a. If the exchange rate is determined by the equality of supply and demand for foreign exchange and the central bank makes no...
-
A local dealer sells baseballs for $25.00 each. If this is a 76.7% markup on cost, find the equivalent markup percent on the selling price
-
An item of depreciable machinery was acquired on 1 July 2009 for $120,000 by cash It is expected to have a useful life of 10 years and zero salvage value On 1 July 2012, it was decided to revalue the...
-
The decomposition of hydrogen peroxide was studied at a particular temperature. The following data were obtained, where Determine the integrated rate law, the differential rate law, and the value of...
-
The lattice energies of FeCl3, FeCl2, and Fe2O3 are (in no particular order) 2631 kJ/ mol, 5339 kJ/ mol, and 14,774 kJ/ mol. Match the appropriate formula to each lattice energy.
-
Consider the following computer- generated model of caffeine. Draw a Lewis structure for caffeine in which all atoms have a formal charge of zero. HONC
-
The suspension system of a car traveling on a bumpy road has a stiffness of \(k=5 \times 10^{6} \mathrm{~N} / \mathrm{m}\) and the effective mass of the car on the suspension is \(m=750...
-
The history of statistical hypothesis testing really began with a tea-tasting experiment (Fisher, 1935), so it seems fitting for this book to end with one. (This is also a fitting ending because...
-
In Laplace domain, \(\lim _{s ightarrow 0}[s X(s)]\) gives the: a. initial value b. transient value c. steady-state value

Study smarter with the SolutionInn App


