Sodium sulfate, Na2SO4 with a mass of 15 g is dissolved in 250 g water. What...
Fantastic news! We've Found the answer you've been seeking!
Question:
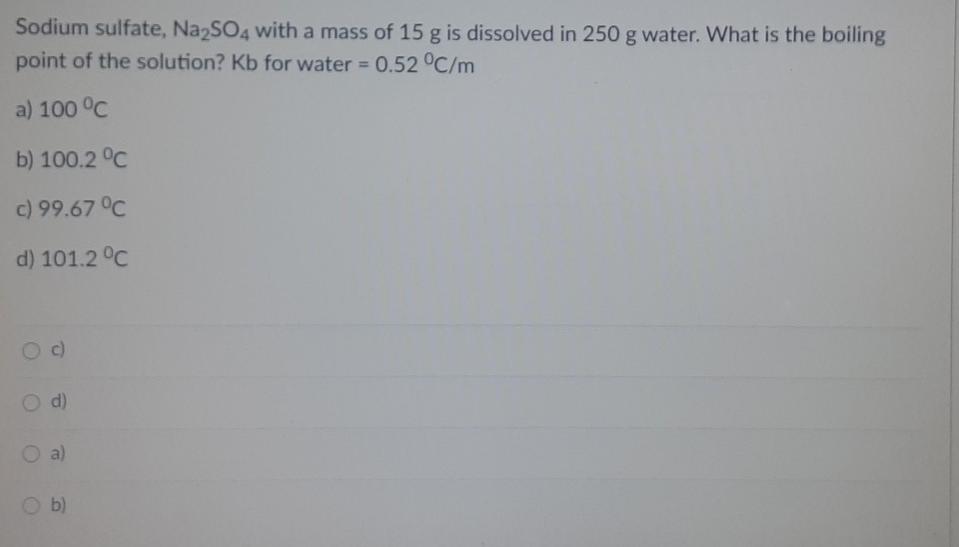


Transcribed Image Text:
Sodium sulfate, Na2SO4 with a mass of 15 g is dissolved in 250 g water. What is the boiling point of the solution? Kb for water = 0.52 °C/m a) 100 °C b) 100.2 °C c) 99.67 °C d) 101.2 °C c) d) O a) b) Fifty grams of an unknown nonelectrolyte solute is mixed with 2000 g of naphthalene (C10H8). The boiling point of the solution was observed to be equal to 80°C. If the boiling point elevation constant of naphthalene is 6.9 °C/m and the boiling point of pure naphthalene is 74.6°C: What is the boiling point elevation of the solution? a) 4 °C b) 5.0 °C c) 5.40 °C d) 5.50 °C a) b) Od) Fifty grams of an unknown nonelectrolyte solute is mixed with 2000 g of naphthalene (C10H8). The boiling point of the solution was observed to be equal to 80°C. If the boiling point elevation constant of naphthalene is 6.9 °C/m and the boiling point of pure naphthalene is 74.6°C: Compute for the molality of the solution. 0.0783 m O 0.783 m 0.0078 m 0.0782 m Sodium sulfate, Na2SO4 with a mass of 15 g is dissolved in 250 g water. What is the boiling point of the solution? Kb for water = 0.52 °C/m a) 100 °C b) 100.2 °C c) 99.67 °C d) 101.2 °C c) d) O a) b) Fifty grams of an unknown nonelectrolyte solute is mixed with 2000 g of naphthalene (C10H8). The boiling point of the solution was observed to be equal to 80°C. If the boiling point elevation constant of naphthalene is 6.9 °C/m and the boiling point of pure naphthalene is 74.6°C: What is the boiling point elevation of the solution? a) 4 °C b) 5.0 °C c) 5.40 °C d) 5.50 °C a) b) Od) Fifty grams of an unknown nonelectrolyte solute is mixed with 2000 g of naphthalene (C10H8). The boiling point of the solution was observed to be equal to 80°C. If the boiling point elevation constant of naphthalene is 6.9 °C/m and the boiling point of pure naphthalene is 74.6°C: Compute for the molality of the solution. 0.0783 m O 0.783 m 0.0078 m 0.0782 m
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the boiling point of a solution of 0.150 g of glycerol, C3H8O3, in 20.0 g of water? What is the freezing point?
-
What is the boiling point of a solution made by adding 6.69 g of magnesium chloride to 243.3 g of water? Use the formula of the salt to obtain i.
-
A railroad car having a mass of 15 Mg is coasting at 1.5 m/s on a horizontal track. At the same time another car having a mass of 12 Mg is coasting at 0.75 m/s in the opposite direction. It the cars...
-
Consider the following graph. 2 7 6 C 10 10 H 11 E 12 15 14 (a). Find a minimum spanning tree of the graph using Kruskal's algorithm. List the edges in the order they are put into the tree. (b)....
-
An average worker in Brazil can produce an ounce of soybeans in 20 minutes and an ounce of coffee in 60 minutes, while an average worker in Peru can produce an ounce of soybeans in 50 minutes and an...
-
In Problem P13.4, if the destination node is located at 5 hops apart from a given source node, what is the maximum possible value of (a) The number of alternate paths of length of 5 hops? (b)...
-
The accounts of Highland Consulting, Inc., follow with their normal balances at August 31, 2010. The accounts are listed in no particular order. Requirements 1. Prepare the companys trial balance at...
-
The president of Healthy Heart Exercise Equipment Co. has been concerned about the growth in costs over the last several years. The president asked the controller to perform an activity analysis to...
-
f(x,y)=2xy-1/2(x^4-y^4)+1 Find all the critical points of f(x,y). After finding them, can you give the function value at each critical point.
-
A textile company produces shirts and pants. Each shirt requires two square yards of cloth, and each pair of pants requires three square yards of cloth. During the next two months the following...
-
Both the latent heat of melting (heat of fusion) and the latent heat of freezing involve the absorption or release of ________ calories per gram of water changing state. a. 100 b. 80 c. 585 d. 540
-
14. Evaluate log 16
-
LenDin company purchases for s inventory are as follows: 20% in the month of purchase, 50% in the following month, and 30% in the second month following purchase. Budgeted inventory purchases for...
-
Bain & Company invented the Net Promoter System, and this system continues to be the leader in how companies earn customer loyalty and inspire employees. What is the Net Promoter Score? How is it...
-
What other expenses might be commonly forgotten when considering the costs of an expansion decision(for example, trash removal, water)? Would these expenses make a material difference in the...
-
Assume that your single aunt Zelda becomes very ill and makes a gift of $ 1 3 , 0 0 0 , 0 0 0 to you. Assuming that she has not made any previous taxable gifts, calculate the gift tax that she will...
-
Jermaine has gotten his dream job working with one of the most prominent investment companies on Wall Street, and he is determined to make a good showing from the first day. What is the best way for...
-
When an electric field is applied to a shallow bath of vegetable oil, why do tiny bits of thread floating in the oil align with the field like compasses in a magnetic field?
-
What is wrong with the following "proof" that every matrix with at least two rows is row equivalent to a matrix with a zero row? Perform R2 + R1 and R1 + R2. Now rows 1 and 2 are identical. Now...
-
Suppose that A is a 6 6 matrix with characteristic polynomial cA() = (1 + ) (1 - )2 (2 - )3. a. Prove that it is not possible to find three linearly independent vectors v1, v2, v3 in R6 such that...
-
In Exercises 1-3, a matrix A is given along with an iterate x5, produced as in Example 4.30. (a) Use these data to approximate a dominant eigenvector whose first component is 1 and a corresponding...
-
Nautical Marina needs to raise \(\$ 1.0\) million to expand the company. Nautical Marina is considering the issuance of either - \(\$ 1,000,000\) of \(8 \%\) bonds payable, or - 100,000 common shares...
-
The board of directors of Circuits Plus authorizes the issue of \(\$ 9,000,000\) of \(8 \%\). 25 -year bonds payable. The semiannual interest dates are May 31 and November 30. The bonds are issued on...
-
The following transactions of Smooth Sounds Music Company occurred during 2020 and 2021 : {Requirement} Record the transactions in Smooth Sounds's journal. Explanations are not required. 2020 Mar. 3...

Study smarter with the SolutionInn App



