In 2009, it was reported that a Mo-Mo quintuple bond (10, 2T, and 25 bonds) could...
Fantastic news! We've Found the answer you've been seeking!
Question:
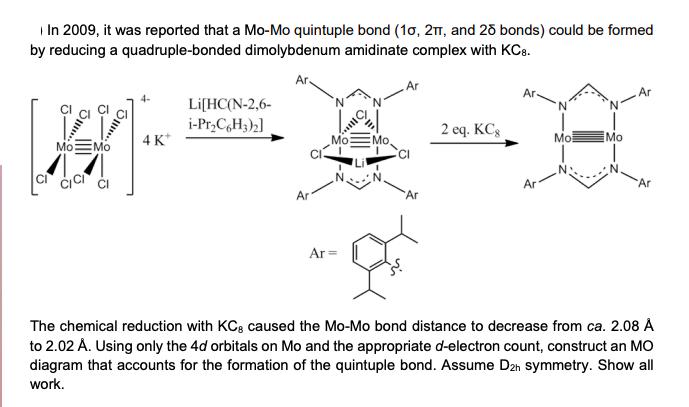
Transcribed Image Text:
In 2009, it was reported that a Mo-Mo quintuple bond (10, 2T, and 25 bonds) could be formed by reducing a quadruple-bonded dimolybdenum amidinate complex with KC8. Ar. Ar Ar- 4- Li[HC(N-2, 6- i-Pr₂C6H3)2] 2 eq. KCg Mo Mo 4K* Mo Mo. Mo: Mo Ar Ar Ar Ar= Ar Ar The chemical reduction with KCg caused the Mo-Mo bond distance to decrease from ca. 2.08 Å to 2.02 A. Using only the 4d orbitals on Mo and the appropriate d-electron count, construct an MO diagram that accounts for the formation of the quintuple bond. Assume D2h symmetry. Show all work. In 2009, it was reported that a Mo-Mo quintuple bond (10, 2T, and 25 bonds) could be formed by reducing a quadruple-bonded dimolybdenum amidinate complex with KC8. Ar. Ar Ar- 4- Li[HC(N-2, 6- i-Pr₂C6H3)2] 2 eq. KCg Mo Mo 4K* Mo Mo. Mo: Mo Ar Ar Ar Ar= Ar Ar The chemical reduction with KCg caused the Mo-Mo bond distance to decrease from ca. 2.08 Å to 2.02 A. Using only the 4d orbitals on Mo and the appropriate d-electron count, construct an MO diagram that accounts for the formation of the quintuple bond. Assume D2h symmetry. Show all work.
Expert Answer:
Answer rating: 100% (QA)
Here is the stepbystep working to construct the MO diagram for the MoMo quintuple bond 1 We a... View the full answer

Related Book For 

Management Accounting Information for Decision-Making and Strategy Execution
ISBN: 978-0137024971
6th Edition
Authors: Anthony A. Atkinson, Robert S. Kaplan, Ella Mae Matsumura, S. Mark Young
Posted Date:
Students also viewed these accounting questions
-
Write in C++ Task 1 and 2 have been completed. Just need Tasks 3 and 4 Introduction The Caesar cipher is a substitution cipher where each letter in the original message (called the plaintext) is...
-
Diego Company manufactures one product that is sold for $80 per unit in two geographic regions-the East and West regions. The following information pertains to the company's first year of operations...
-
Haas Company manufactures and sells one product. The following information pertains to each of the company's first three years of operations: Variable costs per unit: Manufacturing: Direct materials...
-
The distance between the K+ and Cl ions in KCl is 2.80 1010 m. Calculate the energy required to separate the two ions to an infinite distance apart, assuming them to be point charges initially at...
-
What diene and what dienophile should be used to synthesize the following compounds? a. b. c. d. e. f. 0 2 OCH COCH COOH COOH
-
Express in terms of sin only: a. cos 2 b. tan 2 c. cos tan d. cos /tan e. (cos sin )(cos + sin )
-
When performing trend analysis, each line item is expressed as a percentage of: a. net income. b. the base year amount. c. the prior year amount. d. total assets.
-
On October 5, Narveson Company buys merchandise on account from Rossi Company. The selling price of the goods is $5,000, and the cost to Rossi Company is $3,000. On October 8, Narveson returns...
-
Which part of an asset is based on an estimate? cost of getting the asset ready for use O cost of the asset O pattern of use Both A and B
-
You are working on a free-form Packet Tracer challenge activity as seen in Figure 1, you have been given the London Railways network.' The purpose of this EMA question is to build upon each of the...
-
Explain what Du Bois means by "double consciousness" and describe examples (from your life or from Chesnutt's The Marrow of Tradition) Du Bois's The Souls of Black Folk speech
-
Calculate the expected return of GM's stock the using the Capital Asset Pricing Model. Use 1% as the risk- free rate, 1.3 as , and 5% as the expected market rate. GM's Expected Return using the...
-
Given f"(x) = -4 sin(2x) and f'(0) = 2 and f(0) = -5. T Find f 5 ( 1}) =
-
1. Present the information collected systematically, according to the methodology used (2 points). 2. Develop an analysis of the systematized information, highlighting the main elements detected in...
-
Estimate the annual growth rate in PepsiCo's dividends over the 2008-2013 period using the data given in the case. Calculate both the average annual growth rates in PepsiCo's dividends over the...
-
This is the original Sales growth is expected to grow by .0348 or 3.48% (given). From the assumptions, one learns that the firm is at 100% capacity. Therefore all assets are assumed to be...
-
The George's have a balance of $7458.32 in their check register. On their bank statement, the balance on the account is $3365.32. Not reported on the bank statement are checks of $318.65, $29,...
-
Beginning with a country that has a trade deficit, demonstrate graphically what will happen to a countrys potential output with globalization if that countrys costs of production fall. Explain your...
-
Mira Vista Planters provides reforestation services to large paper products companies. It must hire one planter for every 10,000 trees that it has contracted to plant each month. New employees are...
-
What does control mean in a decentralized organization?
-
What are predetermined indirect cost rates commonly called in practice?
-
In 2006, CKX, Inc., a New York sports entertainment company, paid \($50\) million for an 80 percent ownership interest in G.O.A.T. LLC. (G.O.A.T. stands for Greatest of All Time and is a business...
-
In 2006, Danaher, Inc. announced its intent to acquire Sybron Dental Specialties, Inc., a manufacturer of dental products. Danaher indicated that it had agreed to pay \($47\) per share for all of...
-
Aztar Corporation is an operator of casinos and hotels. The companys flagship properties are the Tropicana Hotel & Casino located in Las Vegas, Nevada, and in Atlantic City, New Jersey. In early...

Study smarter with the SolutionInn App


