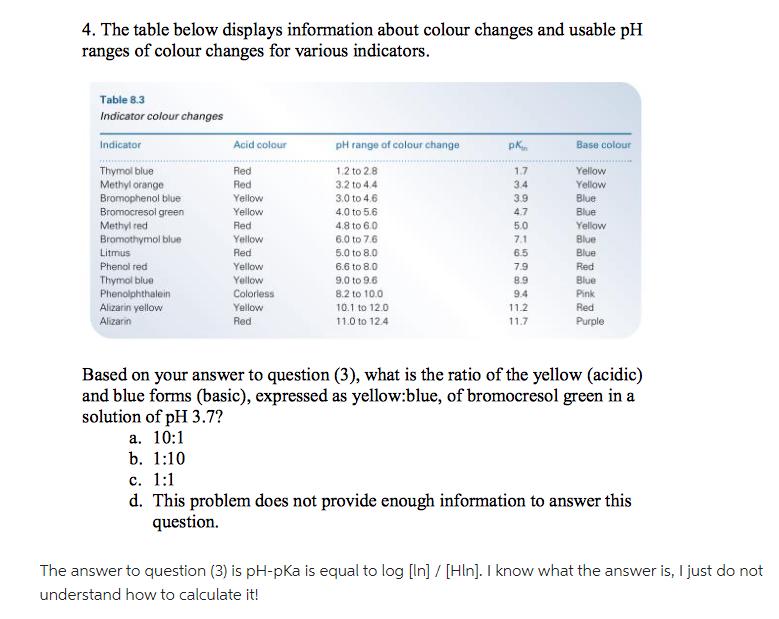
The table below displays information about colour changes and usable pH ranges of colour changes for...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The table below displays information about colour changes and usable pH ranges of colour changes for various indicators. Table 8.3 Indicator colour changes Indicator Thymol blue Methyl orange Bromophenol blue Bromocresol green Methyl red Bromothymol blue Litmus Phenol red Thymol blue Phenolphthalein Alizarin yellow Alizarin Acid colour Red Red Yellow Yellow Red Yellow Red Yellow Yellow Colorless Yellow Red pH range of colour change 1,2 to 2.8 3.2 to 4.4 3.0 to 4.6 4.0 to 5.6 4.8 to 6.0 6.0 to 7.6 5.0 to 8.0 6.6 to 8.0 9.0 to 9.6 8.2 to 10.0 10.1 to 12.0 11.0 to 12.4 pk 1.7 3.4 3.9 4.7 5.0 7.1 76 6.5 7.9 8.9 9.4 11.2 11.7 Base colour Yellow Yellow Blue Blue Yellow Blue Blue Red Blue Pink Red Purple Based on your answer to question (3), what is the ratio of the yellow (acidic) and blue forms (basic), expressed as yellow:blue, of bromocresol green in a solution of pH 3.7? a. 10:1 b. 1:10 c. 1:1 d. This problem does not provide enough information to answer this question. The answer to question (3) is pH-pKa is equal to log [In] / [Hin]. I know what the answer is, I just do not understand how to calculate it! The table below displays information about colour changes and usable pH ranges of colour changes for various indicators. Table 8.3 Indicator colour changes Indicator Thymol blue Methyl orange Bromophenol blue Bromocresol green Methyl red Bromothymol blue Litmus Phenol red Thymol blue Phenolphthalein Alizarin yellow Alizarin Acid colour Red Red Yellow Yellow Red Yellow Red Yellow Yellow Colorless Yellow Red pH range of colour change 1,2 to 2.8 3.2 to 4.4 3.0 to 4.6 4.0 to 5.6 4.8 to 6.0 6.0 to 7.6 5.0 to 8.0 6.6 to 8.0 9.0 to 9.6 8.2 to 10.0 10.1 to 12.0 11.0 to 12.4 pk 1.7 3.4 3.9 4.7 5.0 7.1 76 6.5 7.9 8.9 9.4 11.2 11.7 Base colour Yellow Yellow Blue Blue Yellow Blue Blue Red Blue Pink Red Purple Based on your answer to question (3), what is the ratio of the yellow (acidic) and blue forms (basic), expressed as yellow:blue, of bromocresol green in a solution of pH 3.7? a. 10:1 b. 1:10 c. 1:1 d. This problem does not provide enough information to answer this question. The answer to question (3) is pH-pKa is equal to log [In] / [Hin]. I know what the answer is, I just do not understand how to calculate it!
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Based on your answer to question 4, should Mesa consider hedging its exchange rate risk? If so, explain how it could hedge using forward contracts, futures contracts, and currency options.
-
Based on your answer to Question 5-17, if Pennsylvania's government aims to correct the steel market's negative externality via an effluent fee, is the appropriate fee higher or lower now that steel...
-
Based on your answer to question 9, construct a first-quarter 2015cash flow forecast for Westmark Industrial. Westmark Industrials annual income statement and balance sheet for December 31, 2014...
-
Show that in the limit ??x ?? 0 and ??t ?? 0, the difference Equation (3.12) is equivalent to the differential Equation (2.5). GIVENThe difference equation for one-dimensional transient...
-
In this simulation, you will address questions related to inventory valuation andmeasurement. KWW Professlonal Simulation Inventory Valuation Time Remaining hour 40 minutes Unsplit Split Horie Spit...
-
Discuss the advantages and disadvantages of leasing a car?
-
Marie Janes encounters the following situations in doing the audit of a large auto dealership. Janes is not a partner. 1.The sales manager tells her that there is a sale on new cars (at a substantial...
-
A committee in charge of promoting a Ladies Professional Golf Association tournament is trying to determine how best to advertise the event during the two weeks prior to the tournament. The committee...
-
Read and analyze each item carefully then calculate what is being asked. Show your complete solutions in a good accounting form. . 4. What is the amount of checks outstanding can June 30? * 5 point...
-
Suppose Ann is working on a project with John. Both must decide whether to put into a decent amount of effort into the project. Since Ann is the leader of the project, her contribution determines...
-
4. Products A, B, C, and D have contribution margins of $2.00, $3.00, $4.00, and $5.00, respectively, and require 1.5, 2, 2.5, and 3 machine hours per unit, respectively. Assuming that all units...
-
The emergence of the COVID-19 pandemic in the United States in 2020 signaled the beginning of a twofold economic and public health crisis. The pandemic's aftermath was managed by a patchwork of...
-
Compare in detail and contrast the just in time concept with the focused factory concept in terms of their applicability to a healthcare operational setting.
-
You have been given the following investment decision-making matrix: States of Nature Security Growth Medium Growth No Change Low Bonds 15% 11% 10% 6% Stocks 22% 14% 8% -2% Deposit 10% 10% 10% 10% i)...
-
Explain the "elements of value" concept and note how firms use this concept to increase revenue. Provide a detail answer.
-
. HEMI purchased land and a building for $360,000,000 on September 1, 2023. The land was appraised at $100,000,000, and the building was appraised at 300,000,000 for insurance purposes. Management...
-
Question 1 The Manufacturing Company has 10 tugger trains, 15 AGVs in its plant. These systems needs to pick and drop the raw material and finished goods with in plant operations, and sometime...
-
One Way Cellular accountants have assembled the following data for the year ended September 30, 2014: Prepare the operating activities section using the indirect method for One Way Cellulars...
-
As the inside radius of an open tubular column is decreased, the maximum possible column efficiency increases and sample capacity decreases. For a thin stationary phase that equilibrates rapidly with...
-
Fill in the blanks: (a) The product of a reaction between a Lewis acid and a Lewis base is called ________. (b) The bond between a Lewis acid and a Lewis base is called ________ or _______. (c)...
-
Combustion analysis of an organic compound gave the composition 71.17 0.41 wt% C, 6.76 0.12 wt% H, and 10.34 0.08 wt% N. Find the coefficients h and n and their uncertainties x and y in the...
-
1. Benzaquin had a radio talk show in Boston. On the program, he complained about an incident earlier in the day, in which state trooper Fleming had stopped his car, apparently for lack of a proper...
-
3. Fifteen-year-old Terri Stubblefield was riding in the back seat of a Ford Mustang II when the car was hit from behind. The Mustang was engulfed in a ball of fire, and Terri was severely burned....
-
2. Caldwell, carrying a large purse, was shopping in a K-Mart store. A security guard observed her look at various small items such as stain, hinges, and antenna wire. On occasion she bent down out...

Study smarter with the SolutionInn App



