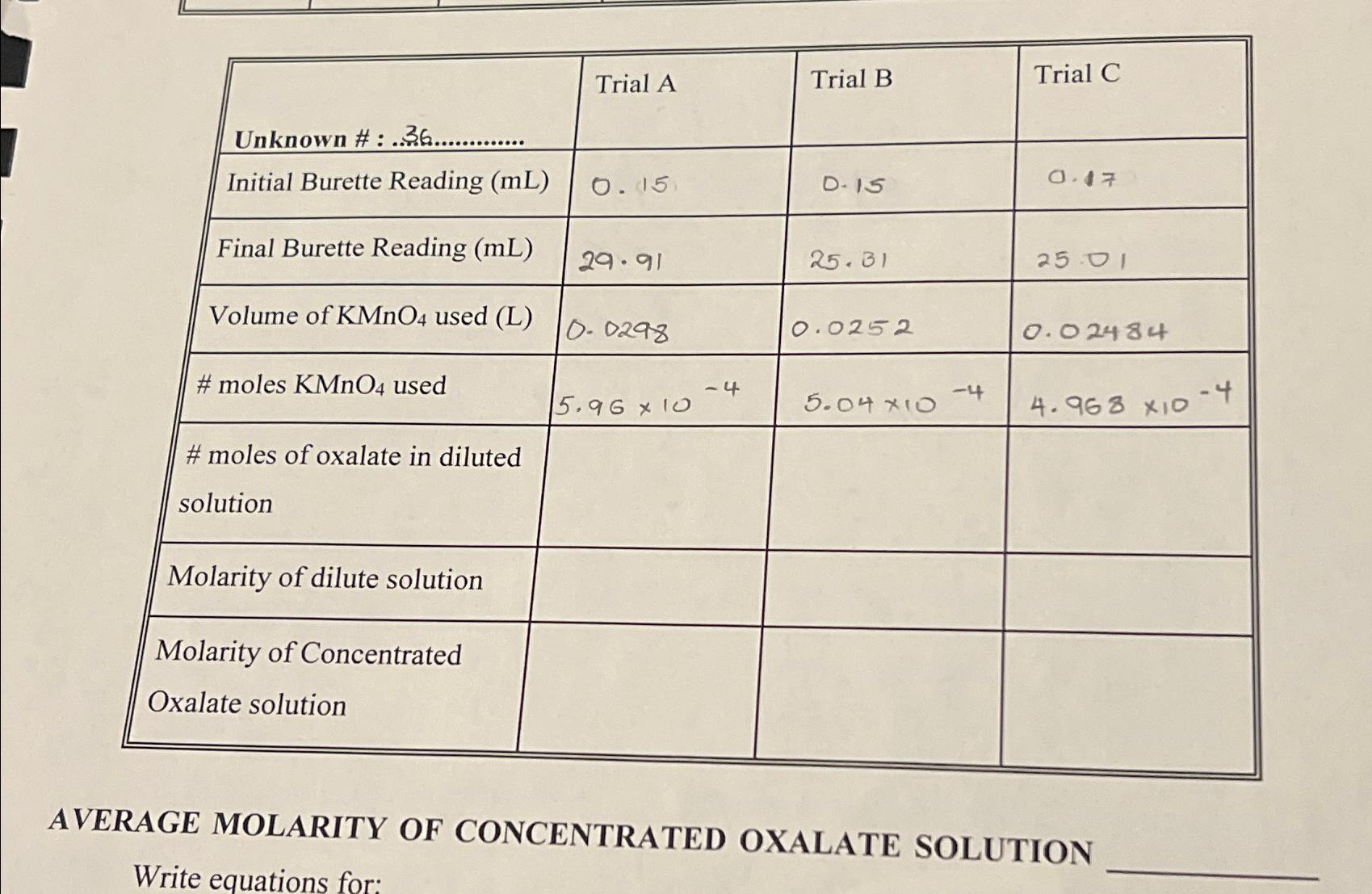
Unknown #:.36............. Initial Burette Reading (mL) Final Burette Reading (mL) Volume of KMnO4 used (L) #moles...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Unknown #:.36............. Initial Burette Reading (mL) Final Burette Reading (mL) Volume of KMnO4 used (L) #moles KMnO4 used #moles of oxalate in diluted solution Molarity of dilute solution Molarity of Concentrated Oxalate solution Trial A 0.15 29.91 0.0298 5.96 x 10 -4 Trial B D-15 25.31 0.0252 5.04 *10 -4 Trial C 0.47 25.01 0.02484 4.968 X10 AVERAGE MOLARITY OF CONCENTRATED OXALATE SOLUTION Write equations for: -4 Unknown #:.36............. Initial Burette Reading (mL) Final Burette Reading (mL) Volume of KMnO4 used (L) #moles KMnO4 used #moles of oxalate in diluted solution Molarity of dilute solution Molarity of Concentrated Oxalate solution Trial A 0.15 29.91 0.0298 5.96 x 10 -4 Trial B D-15 25.31 0.0252 5.04 *10 -4 Trial C 0.47 25.01 0.02484 4.968 X10 AVERAGE MOLARITY OF CONCENTRATED OXALATE SOLUTION Write equations for: -4
Expert Answer:
Answer rating: 100% (QA)
Balanced ionic reaction betweeen oxalate and KMnO4 is VII 2 5 C0 2 MnO 16H moles of KMnO ... View the full answer

Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Posted Date:
Students also viewed these physics questions
-
The stock of Marvin Gardens currently sells for $133.75 per share. The firm has a constant dividend growth rate of 2%. If the required rate of return is 12%, what will the stock sell for five years...
-
A road building company is purchasing an excavator for $500,000. The corporate tax rate is 45% and the expected after-tax rate of return is 12%. What would be the present value of the future tax...
-
A survey of information systems managers was used to predict the yearly salary of beginning programmer/analysts in a metropolitan area. Managers specified their standard salary for a beginning...
-
According to adaptive expectations theory, a decline in the short-run aggregate demand curve ____ the inflation rate and ____ the unemployment rate. a) decreases; increases b) increases; decreases c)...
-
Mrs. Edwards presently operates a retailing proprietorship with a December 31 fiscal year-end. She expects net income for tax purposes of $210,000 from the business. Since the business currently...
-
Rod AB is held in place by the cord AC. Knowing that the tension in the cord is 1350 N and that c = 360 mm, determine the moment about B of the force exerted by the cord at point A by resolving that...
-
Continuation of P10-35B: cash budgets (Learning Objective 3) Refer to P10-35B. Go Sports sales are 50% cash and 50% credit (use sales on the last two lines of P10-35B). Credit sales are collected in...
-
Common- size and trend percents for Rustynail Companys sales, cost of goods sold, and expenses follow. Determine whether net income increased, decreased, or remained unchanged in this three-...
-
16) Calculate the limits algebraically. Provide an argument justifying the value stated: a) lim(x - vx2 -5x) X->00 3x + 5sin x b) lim x->0 2x - cosx20) Your task is to build a road joining a ranch to...
-
We have a digital medium with a data rate of 10 Mbps. How many 64-kbps voice channels can be carried by this medium if we use DSSS with the Barker sequence?
-
A comparative income statement is given below for McKenzie Sales, Ltd., of Toronto: McKenzie Sales, Ltd. Comparative Income Statement Sales Cost of goods sold This Year Last Year $7,340,000...
-
Briefly describe a job you have or had. (If you havent had a job, think of someone you know well.) If you were to be promoted, which training method(s) would you use to train the person to do your...
-
You are the external auditor for Centnet Pty. Ltd., a public electronic funds transfer network that operates switches in the capital cities of all states in Australia. Because Centnet has a large...
-
To provide more extensive services to their customers, financial institutions in some countries are becoming increasingly involved in using interchange network facilities. In an interchange network...
-
You are the partner-in-charge of information systems auditing for a large public accounting firm. One of your clients is a major insurance company that is a mature user of computer-based information...
-
During a professional development program at a CPA firm, an audit manager was instructing several younger staff members on how to calculate confidence intervals in the accounts receivable area by...
-
ces At the beginning of June, Circuit Country has a balance in inventory of $2,650. The following transactions occur during the month of June. June 2 Purchase radios on account from Radio World for...
-
One Way Cellular accountants have assembled the following data for the year ended September 30, 2014: Prepare the operating activities section using the indirect method for One Way Cellulars...
-
One kilogram of high-grade coal produces about 2.8 10 4 kJ energy when it is burned. Fission of 1 mol 235 U releases 1.9 10 10 kJ. (a) Calculate the number of metric tons (1 metric ton = 1000 kg)...
-
The accepted atomic mass of nitrogen is 14.0067 u. Approximately 99.632% of natural nitrogen is 14 N, which has an isotopic mass of 14.0031 u. Th e remaining nitrogen is 15 N. What is the isotopic...
-
Complete and balance the following oxidation-reduction reactions. Strategy Use the half-reaction method to balance redox reactions.
-
Karanga Company purchased land containing an estimated 20 million tons of ore for a cost of $6,600,000. The land without the ore is estimated to be worth $1,200,000. The company expects that all the...
-
The airline industry was hit particularly hard after the 9/11 attacks on the World Trade Center in 2001. In 2002, Southwest Airlines, one of the healthier airlines companies, made a decision to...
-
Gent Manufacturing Company purchased a robot for $720,000 at the beginning of year 1. The robot has an estimated useful life of four years and an estimated residual value of $60,000. The robot, which...

Study smarter with the SolutionInn App


