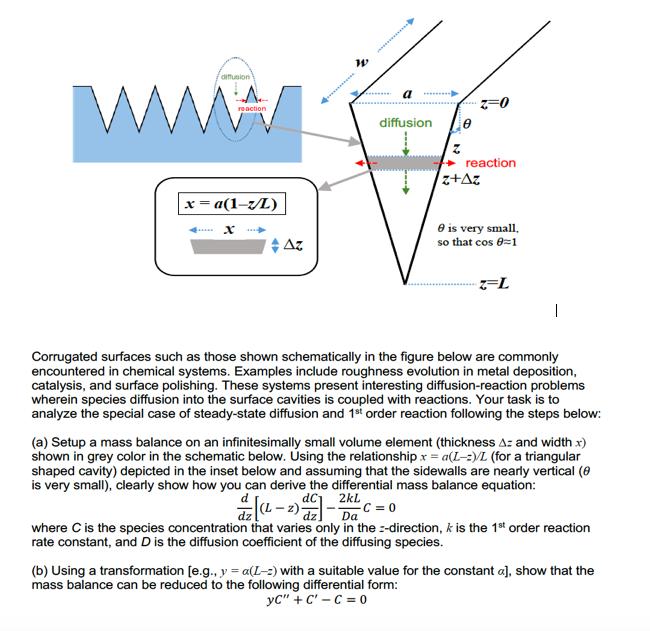
W diffusion reaction x=a(1-z/L) x Az W a diffusion Z=0 reaction. z+Az 8 is very small,...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
W diffusion reaction x=a(1-z/L) x Az W a diffusion Z=0 reaction. z+Az 8 is very small, so that cos 8=1 z-L Corrugated surfaces such as those shown schematically in the figure below are commonly encountered in chemical systems. Examples include roughness evolution in metal deposition, catalysis, and surface polishing. These systems present interesting diffusion-reaction problems wherein species diffusion into the surface cavities is coupled with reactions. Your task is to analyze the special case of steady-state diffusion and 1st order reaction following the steps below: (a) Setup a mass balance on an infinitesimally small volume element (thickness A: and width x) shown in grey color in the schematic below. Using the relationship x = a(L-:)/L (for a triangular shaped cavity) depicted in the inset below and assuming that the sidewalls are nearly vertical (8 is very small), clearly show how you can derive the differential mass balance equation: dC 2kL (1-2) - Da -C=0 where C is the species concentration that varies only in the direction, k is the 1st order reaction rate constant, and D is the diffusion coefficient of the diffusing species. (b) Using a transformation [e.g., y = a(L-2) with a suitable value for the constant a], show that the mass balance can be reduced to the following differential form: yC" + C-C=0 W diffusion reaction x=a(1-z/L) x Az W a diffusion Z=0 reaction. z+Az 8 is very small, so that cos 8=1 z-L Corrugated surfaces such as those shown schematically in the figure below are commonly encountered in chemical systems. Examples include roughness evolution in metal deposition, catalysis, and surface polishing. These systems present interesting diffusion-reaction problems wherein species diffusion into the surface cavities is coupled with reactions. Your task is to analyze the special case of steady-state diffusion and 1st order reaction following the steps below: (a) Setup a mass balance on an infinitesimally small volume element (thickness A: and width x) shown in grey color in the schematic below. Using the relationship x = a(L-:)/L (for a triangular shaped cavity) depicted in the inset below and assuming that the sidewalls are nearly vertical (8 is very small), clearly show how you can derive the differential mass balance equation: dC 2kL (1-2) - Da -C=0 where C is the species concentration that varies only in the direction, k is the 1st order reaction rate constant, and D is the diffusion coefficient of the diffusing species. (b) Using a transformation [e.g., y = a(L-2) with a suitable value for the constant a], show that the mass balance can be reduced to the following differential form: yC" + C-C=0
Expert Answer:
Answer rating: 100% (QA)
2 X1 X1 addeve To be designed using number of halbad... View the full answer

Related Book For 

Fundamentals Of Momentum Heat And Mass Transfer
ISBN: 9781118947463
6th Edition
Authors: James Welty, Gregory L. Rorrer, David G. Foster
Posted Date:
Students also viewed these chemical engineering questions
-
In Graph 9.4, we illustrated how you can derive the labor supply curve from a consumer model in which workers choose between leisure and consumption. A: In end-of-chapter exercise 3.1, you were asked...
-
Shown schematically in the figure below is a thin and laterally large conducting plate. This plate is charged with the net charge density per unit area o=-16.74 nC/m2 thereby becoming the source of...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
The following data represent the muzzle velocity (in feet per second) of rounds red from a 155-mm gun. For each round, two measurements of the velocity were recorded using two different measuring...
-
How important is product design in life cycle costing? Why?
-
Identify the accounts affected by each of the following transactions, and state whether each account is debited or credited: Income Statement Balance Sheet Account (Dr/Cr) Account (Dr/Cr) Transaction...
-
Jack Swanson gave a creditor a 90-day, eight percent note payable for \(\$ 7,200\) on December 16 . What adjusting entry should Swanson make on December 31 ?
-
Stark Company has five employees. Employees paid by the hour receive a $10 per hour pay rate for the regular 40-hour workweek plus one and one-half times the hourly rate for each overtime hour beyond...
-
A $5300 loan at 6.6% compounded monthly was settled by a single payment of $6500 including accrued interest. Rounded to the nearest day, how long after the initial loan was the $6500 payment made?...
-
Required Tasks: 1. Compute the probability that the sampling plan will provide a result that suggests that SaveMor should reject the deal even if the true proportion of all customers who would switch...
-
2) For the scenarios below, identify an audit procedure that you would perform to address the audit risk within the scenario and which account/assertion the audit procedure is addressing. Scenario...
-
2. The normal melting point of mercury is -38.87C. At this temperature, the density of liquid mercury is 13.65 g/cm and the density of solid mercury is 14.26 g/cm3. The heat of fusion is 2.333...
-
what was the AMERICAN REVOLUTION and what were three causes of the American Revolution? The Latin American Revolutions and the Haitian Revolution were much more radical than the American Revolution...
-
Consider the following function and then answer the question 19 and 20. ALGORITHM q19p (A [1...r]) BEGIN p=A[1]; i=1;j=r+1 repeat repeat i=i+1 until A[i]>=p repeat j-j-1 until A[j] =j swap (A[i],...
-
[The following information applies to the questions displayed below.] Shauna Coleman is single. She is employed as an architectural designer for Streamline Design (SD). Shauna wanted to determine her...
-
Gladstorm Enterprises sells a product for $51 per unit. The variable cost is $38 per unit, while fixed costs are $4,485. Determine the: Round to the nearest whole number of units. a. Break-even point...
-
7. The US-government under George W. Bush put on September 7, 2008, Fannie Mae, and Freddie Mac under the conservatorship of the FHFA (i.e., the government took over both institutions). Please...
-
Presented below are income statements prepared on a LIFO and FIFO basis for Kenseth Company, which started operations on January 1, 2024. The company presently uses the LIFO method of pricing its...
-
A spherical silica gel adsorbent particle of 0.20 cm diameter in placed in a rapidly flowing air stream at a velocity of 50 cm/s containing 1.0 mole% of H 2 O vapor at total system pressure of 1.0...
-
A furnace wall consisting of 0.25 m of fire clay brick, 0.20 m of kaolin, and a 0.10-m outer layer of masonry brick is exposed to furnace gas at 1370 K with air at 300 K adjacent to the outside wall....
-
Wastewater contaminated with 1,2,2-trichioroethane (C 2 H 3 C1 3 , molecular weight 133.5 g/gmole) will be treated in a counter current packed tower using contaminant-free air as the stripping gas....
-
Browne \& Company has no-par value common stock outstanding that is selling at \(\$ 40\) per share. The company's CEO believes that the stock price is undervalued and decides to buy back 10,000...
-
Calculate the dividend yield for Evans \& Sons for 2018 and 2019. Did the dividend yield improve from 2018 to 2019? Evans \& Sons, Inc., disclosed the following information in a recent annual report:...
-
Calculate the dividend payout for Evans \& Sons for 2018 and 2019. Did the dividend payout increase from 2018 to 2019 ? Evans \& Sons, Inc., disclosed the following information in a recent annual...

Study smarter with the SolutionInn App


