Which of the following is correct regarding the pH scale? A substance with a pH of...
Fantastic news! We've Found the answer you've been seeking!
Question:
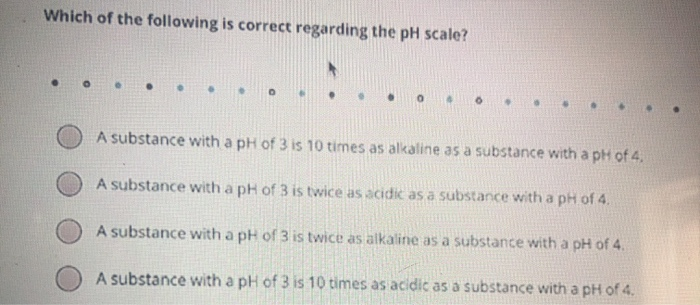
Transcribed Image Text:
Which of the following is correct regarding the pH scale? A substance with a pH of 3 is 10 times as alkaline as a substance with a pH of 4. A substance with a pH of 3 is twice as acidic as a substance with a pH of 4. substance with a pH of 3 is twice as alkaline as a substance with a pH of 4. A substance with a pH of 3 is 10 times as acidic as a substance with a pH of 4. Which of the following is correct regarding the pH scale? A substance with a pH of 3 is 10 times as alkaline as a substance with a pH of 4. A substance with a pH of 3 is twice as acidic as a substance with a pH of 4. substance with a pH of 3 is twice as alkaline as a substance with a pH of 4. A substance with a pH of 3 is 10 times as acidic as a substance with a pH of 4.
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Questio... View the full answer

Related Book For 

Income Tax Fundamentals 2015
ISBN: 9781305177772
33rd Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven Gill
Posted Date:
Students also viewed these accounting questions
-
Which of the following is correct regarding the deduction for self-employment taxes? a. Self-employment taxes are deductible in full to arrive at adjusted gross income b. Self-employment taxes are...
-
Which of the following is correct regarding self-employment taxes? I. For purposes of the portion of self-employment tax that is equivalent to Social Security tax, taxable self-employment income is...
-
Which of the following is correct regarding the secondary source of information? a) Speech b) Government documents c) Articles in journal d) All of the above
-
Plzz solve quicklyy Determine the non-trivial root of: \[ \sin (x)=x^{2} \] Where \( \mathrm{x} \) is in radians. Use the bisection method with initial guesses of \( \mathrm{x}_{0} \mathrm{~m} 0.6 \)...
-
In January 2010, Janas Corporation was organized and authorized to issue 1,000,000 shares of no-par common stock and 25,000 shares of 5 percent, $50 par value, noncumulative preferred stock. The...
-
In Problem 13.10 on page 532, you used box office gross to predict DVD revenue. The data are stored in Movie. The company is about to release a movie on DVD that had a box office gross of $75...
-
The Cunningham Company is in the process of determining a return rate to use for its cost of capital. Upon review of the financial statements it was determined that the total interest bearing debt is...
-
Reconstructing the income statement and balance sheet (Adapted from a problem by Stephen A. Zeff) Portobello Co., a retailer, is in its 10th year of operation. On December 28, 2008, three days before...
-
You want to buy a house that costs $2,000,000. You only have $400,000 that you can spend on the house, so you want to finance the rest with a 30-year fixed-rate mortgage of 3.6% per annum compounding...
-
For 2021, Essence Company, a calendar year taxpayer, will change from the cash method for tax purposes to the accrual method. At the end of 2020, Essence showed the following items. Accounts...
-
Write the process necessary to create a 64-bit arithmetic shift register. In addition to the normal synchronous inputs, your register should also provide the following synchronous shift signals:...
-
You are generating your pro forma income statement for next year. Sales are forecast to be $100,000. Variable operating expenses are expected to be 20% of sales, and fixed operating expenses are...
-
Assume BBB stock price is currently $20. In each six-month period, it will either fall by 50 percent or rise by 100 percent. What is the current value of a one-year call option with an exercise price...
-
A parked police car takes off after a suspect and reaches 78.5 km/h East in 8.20 seconds. What is the average acceleration (m/s^2) of the car? | Select an answer Enter an integer or decimal number,...
-
1. What is the essence of communication for women? How about for men? 2. Describe the masculinity-femininity dimension and offer a specific example not listed in the reading. 3. Why is there a...
-
How can we manage organizational changes effectively. b) How important are managers in the organizational change process.?
-
Based on John Moody's analytics, how can high and low ratings from Moodys affect the interest rate
-
An 8.0 kg crate is pulled 5.0 m up a 30 incline by a rope angled 18 above the incline. The tension in the rope is 120 N, and the crates coefficient of kinetic friction on the incline is 0.25. a. How...
-
In 2014, Gale and Cathy Alexander hosted an exchange student, Axel Muller, for 9 months. Axel was part of International Student Exchange Programs (a qualified organization). Axel attended tenth grade...
-
During 2014, Tom sold Sears stock for $10,000. The stock was purchased 4 years ago for $13,000. Tom also sold Ford Motor Company bonds for $35,000. The bonds were purchased 2 months ago for $30,000....
-
a. Loren is a secretary in a lawyers office. Since he often deals with legal matters, Loren feels that a law degree will be beneficial to him. May Loren deduct his educational expenses for law school...
-
Comparative balance sheets and the intervening statement of income for Sears Canada Inc. appear below. Sears Canada is a subsidiary of Sears Holdings Corporation, a U.S. corporation. All amounts are...
-
On January 1, 2016, the U.K. subsidiary of U.S. International Corporation had the following condensed balance sheet, in pounds sterling (in millions): The exchange rate on January 1, 2016 was...
-
Grupo Comercial Chedraui is a food retailer headquartered in Mexico. While it has several subsidiaries, only one is located outside of Mexico. Bodega Latina Co. is a chain of self-service stores...

Study smarter with the SolutionInn App


