The following reaction begins when 0.56 mol of CO and 0.64 mol of H2O are added...
Fantastic news! We've Found the answer you've been seeking!
Question:
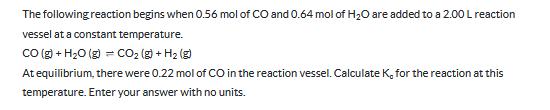
Transcribed Image Text:
The following reaction begins when 0.56 mol of CO and 0.64 mol of H2O are added to a 2.00 L reaction vessel at a constant temperature. CO(g) + H2O (g) = CO2(g) + H2(g) At equilibrium, there were 0.22 mol of CO in the reaction vessel. Calculate K, for the reaction at this temperature. Enter your answer with no units. The following reaction begins when 0.56 mol of CO and 0.64 mol of H2O are added to a 2.00 L reaction vessel at a constant temperature. CO(g) + H2O (g) = CO2(g) + H2(g) At equilibrium, there were 0.22 mol of CO in the reaction vessel. Calculate K, for the reaction at this temperature. Enter your answer with no units.
Expert Answer:
Answer rating: 100% (QA)
The balanced chemical equation for the given reaction is COg H2Og CO2g H2g The reaction quotient Qc ... View the full answer

Related Book For 

Posted Date:
Students also viewed these programming questions
-
A. There are three persons, A, B, and C that work with two ways of painting the building walls. The first way, they separately work alone. "A" is able to complete the task himself in 1 week (6 days),...
-
Assume that you are buying ServiceNow software and deployment services for service desk management. This is for the IT department only, but it affects the whole company in how services from IT will...
-
Big Bite is offering a promotion as follows: If the customer orders $20 or more worth of food, there is a 20% discount If the customer orders between $10 and $20 worth of food, there is a 15%...
-
As a result of the recent mortgage crisis, many banks reported record losses to their mortgage receivables and other assets based on the decline in these assets fair values. Requirements 1. What...
-
A long retaining wall is braced by wood shores set at an angle of 30° and supported by concrete thrust blocks, as shown in the first part of the figure. The shores are evenly spaced, 3 m apart....
-
(a) Why is seniority considered a critical issue? (b) What are the advantages and disadvantages of using a seniority system?
-
Computing unit costs at different levels of production Perfumeria, Inc., budgeted for 12,000 bottles of product Si during the month of May. The unit cost of Si was \(\$ 20\), consisting of direct...
-
Can Jackson delegate her duty to maintain the buildings to Dunn? Why or why not? Who can be held liable for Dunns failure to fix the ceiling, Jackson or Dunn? Was Faught an intended third party...
-
If the spot exchange rate is 124.2 / $, the one-year risk-free rate is 2.1% in U.S. and 1.2% in Japan, what is the one-year forward exchange rate on the yen in yen / $? A. 123.11 / $ B. 124.98 / $ C....
-
Brian Brown, an executive at a manufacturing enterprise, comes to you on December 1 of the current year for tax advice. He has agreed to donate a small tract of land to the Rosepark Community...
-
Thad, a single taxpayer, has taxable income before the QBI deduction of $180, 500. Thad, a CPA, operates an accounting practice as a single member LLC (which he reports as a sole proprietorship)....
-
Helen's husband, Jake, is also considering buying an investment property, but in Adelaide instead. Jake has calculated that he needs to borrow $450,000 from the bank to fund this purchase. The...
-
Orion Ltd. is planning a massive expansion to its manufacturing plant.The cost of the expansion is $10,000,000 which consists ofnew state-of-the-artequipment.The estimated life of the new equipment...
-
What is logistics? And how many types of logistics? And define the example of logistics.
-
Explain briefly the three types of data that is stored in an ERP
-
1. List the types of calls that would be put through immediately and those for which a message should be taken and followed up with a return call. 2. A nurse enter a room and found a patient lying on...
-
On December 31, 2020, Rebel Corporation's balance sheet reported the following. Common stock, $1 par Paid-in capital-excess of par Retained earnings Treasury stock (28,000 shares at cost) $1,000,000...
-
1. Use these cost, revenue, and probability estimates along with the decision tree to identify the best decision strategy for Trendy's Pies. 2. Suppose that Trendy is concerned about her probability...
-
Consider the 2 4 factorial experiment in Example 6-2. Suppose that the last observation is missing. Reanalyze the data and draw conclusions. How do these conclusions compare with those from the...
-
Suppose that in Problem 6-7 ABCD was confounded in replicate I and ABC was con-founded in replicate II. Perform the statistical analysis of this design. Problem 6-7 An experiment was performed to...
-
Consider the three-stage nested design shown in Figure 13-5 to investigate alloy hardness. Using the data that follow, analyze the design, assuming that alloy chemistry and heats are fixed factors...
-
Distinguish between Maximax, Maximin, and Minimax regret decision-making models. Ex- plain why the attitude of the decision maker towards risk taking influences which model he/she would be...
-
Discuss the assumptions underlying probabilistic cost-volume-profit analysis.
-
Explain the reasons why overproducing a good may lead to real costs. How does un- derproducing a good differ from overproducing? Discuss the nature of the costs involved.

Study smarter with the SolutionInn App


