Calculate the number of tetrahedral atoms per nm 3 for each zeolite framework type listed in the
Question:
Calculate the number of tetrahedral atoms per nm3 for each zeolite framework type listed in the table below.
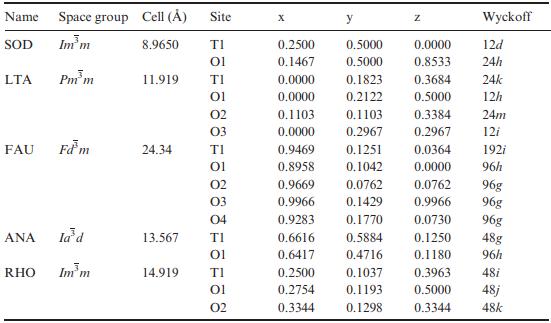
Transcribed Image Text:
Name Space group Cell () SOD Imm LTA FAU ANA RHO Pmm Fdm la'd Im m 8.9650 11.919 24.34 Site TI 01 14.919 TI 01 02 03 TI 01 02 03 04 13.567 TI 01 TI 01 02 X 0.2500 0.5000 0.1467 0.5000 0.0000 0.1823 0.0000 0.2122 0.1103 0.0000 0.9469 0.8958 0.9669 0.9966 0.9283 y 0.2500 0.2754 0.3344 0.1103 0.2967 0.1251 0.1042 0.0762 0.1429 0.1770 0.6616 0.5884 0.6417 0.4716 0.1037 0.1193 0.1298 Z 0.0000 0.8533 0.3684 0.5000 0.3384 0.2967 0.0364 0.0000 0.0762 0.9966 0.0730 0.1250 0.1180 0.3963 0.5000 Wyckoff 12d 24h 24k 12h 24m 12i 192i 96h 96g 96g 96g 48g 96h 48i 48j 0.3344 48k
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
See table Note cells are for the framework type such that T per nm ...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
Zeolite A can be described using a 11.9 primitive cubic cell with the origin at a -cage centerin Figure 14.3. From the figure, make a visual estimate of the size of a sphere that would fit inside...
-
The -cristobalite polymorph of SiO 2 has a tetragonal cell with a = 4.971 , b = 6.922 , with Si on 2a and O on 4f Wyckoff sites. The SiO bond length is 1.60 and the average OO distance within...
-
For the following two graphics, provide the specified information below for each. Inverse Demand: P= 43.75 - .00625 Q; MR = 43.75 - 0.0125 Q 25 20 15 $ per unit 10 10 5 0 MC 500 1000 1500 ATC 2000 -...
-
Use the accompanying graph of y = f(x). Does exist? If it does, what is it? lim f(x)
-
Using the Hund rules, find the magnetic moment of the ground state of the atom whose open sub shell is half-filled with five electrons.
-
Based on demand-control and effort-reward models of stress, are Trader Joes crew members likely to experience a great deal of stress?
-
Matt Enterprises issued \(\$ 200,000\) of ten percent, five-year bonds with interest payable semiannually. Determine the issue price if the bonds are priced to yield (a) ten percent, (b) six percent,...
-
Staci Valek began dabbling in pottery several years ago as a hobby. Her work is quite creative, and it has been so popular with friends and others that she has decided to quit het job is $3,800 per...
-
A new investment opportunity for you is an annuity that pays $500 at the beginning of each year for 3 years. You could earn 5.5% on your money in other investments with equal risk. What is the most...
-
The table above contains ideal framework coordinates for different zeolites. Use a package such as vesta (http://jp-minerals.org/vesta/en/) to produce a 3D drawing of each structure. For each example...
-
Calculate the surface area of (a) A 1 g single crystal of cristobalite (density = 2.3 g/cm 3 , assume the crystal takes a spherical shape), (b) A uniform powder of spherical crystals each with a...
-
The position of the stylus tip A is controlled by the robot shown. In the position shown the stylus moves at a constant speed u = 180mm/s relative to the solenoid BC. At the same time, arm CD rotates...
-
Discuss whether the "Net zero economy target" is a systematic risk factor not included in the current literature.
-
You are advising the owner of a 100k sf warehouse built in 2005 as to whether they should Hold or Sell the property The property is fully occupied by a single tenant on 10yr lease from Jan 2015...
-
Outline what disclosure obligations Aaron and Jacinta are subject to, and why they are important to them as clients?
-
Expain the channels and the dynamics of the effects of leverage cycles on policymakers' responses.
-
Labor's share of total income is: The graphs to the right illustrate the relationship between the market for labor and the aggregate production function for a given economy. According to these...
-
Data from the comparative balance sheet of Sterling Company at March 31, 2012, follow: Sterling Companys transactions during the year ended March 31, 2012, included the following: Requirements 1....
-
The company manufactures three products: wooden chairs, tables and dressers. AFC started off as a 'Mom & Pop' shop but has grown rapidly. AFC uses one assembly line to build all three products,...
-
Predict the final product(s) for each sequence of reactions: (a) (b) (c) (d) Br Br. 1) Excess NaNH, 2) EECI 3) H2, Lindlar's catalyst 1) NANH, 2) Mel 3) 9-BBN 4) H202, NaOH H-CEC-H
-
When (R)-4-bromohept-2-yne is treated with H 2 in the presence of Pt, the product is optically inactive. Yet, when (R)-4-bromohex-2-yne is treated with the same conditions, the product is optically...
-
If the reaction Fe 2 N(s) + 3/2H 2 (g) 2Fe(s) + NH 3 (g) comes to equilibrium at a total pressure of 1 bar, analysis of the gas shows that at 700. and 800. K, PNH 3 /PH 2 = 2.165 and 1.083,...
-
Imagine a firm with the first year FCF of $50M. This firm's FCFs are expected to grow 8% per year until the end of year 4. Starting from year 5, the FCF growth rate will be 2% per year perpetually....
-
Karane Enterprises, a calendar-year manufacturer based in College Station, Texas, began business in 2022. In the process of setting up the business, Karane has acquired various types of assets. Below...
-
In this question you will be calculating two stock portfolio (META and AMZN) and (META and MS) weighted annual returns. Weights are: 100%, 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10% and 0%. Note: if...

Study smarter with the SolutionInn App


