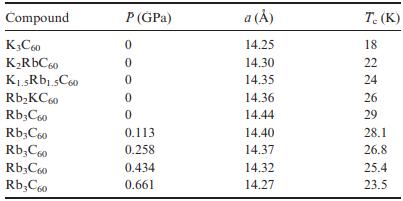
The table below contains data for the alkali-metal fullerides A 3 C 60 . (a) Give a
Question:
The table below contains data for the alkali-metal fullerides A3C60.
(a) Give a brief explanation for the Tc changes observed in the 0 GPa data.
(b) Give a brief explanation for the pressure dependence of Tc in Rb3C60.
(c) Comment on differences in Tc between the two series.
Transcribed Image Text:
Compound K3C60 KRbC60 K.5Rb1.5C60 RbKC60 Rb3C60 Rb3C60 RbC60 Rb3C60 RbC60 P (GPa) 0 0 0 0 0 0.113 0.258 0.434 0.661 a () 14.25 14.30 14.35 14.36 14.44 14.40 14.37 14.32 14.27 T. (K) 18 22 24 26 29 28.1 26.8 25.4 23.5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
abc The data relate to Figure 1210 where the dependence of T c on the volume per C 60 was presented ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
You are currently willing to pay $25,000 to purchase a perpetuity which will pay you and your heirs $2,250 each year forever (the first $2,250 payment will be received one year from today). If your...
-
Watt Wholesale Company purchases merchandise from a variety of manufacturers and sells the merchandise to a variety of retailers. All sales are subject to a cash discount (2/10, n/30). Watt uses a...
-
Discuss whether the following supply of goods and services are taxable events in GST: 1 . A company having warehouse in Mumbai shifted some raw material to its factory in Baroda 2 . An Indian company...
-
Julio sold his corporation to a competitor, Exeter LLC, for $100,000,000. Julio incorporated his business 17 years ago by investing $500,000 plus his proprietary know-how. There have been no other...
-
Find the threshold energy of gamma quantum required to form (a) An electron-positron pair in the field of a stationary electron; (b) A pair of pions of opposite signs in the field of a stationary...
-
If you were to take the team/role-player approach, what steps would you take to help the team members and outsiders deal with the perceived lack of talent?
-
What formula should Hardy Company use to calculate the total amount of interest on a note payable that uses add-on interest?
-
Jones, wishing to retire from a business enterprise that he had been conducting for a number of years, sold all of the assets of the business to Jackson Corp. Included in the assets were a number of...
-
Thaler has studied the impact of the default retirement plan option ( " opt - in " or "opt - out" ) on the decision to enroll in a retirement saving plan. Changing this default option will affect the...
-
Rb x Cs 3x C 60 compounds have T c = 26.9 K, 32.9 K, and 31.8 K and room-temperature cell parameters a = 14.70 , 14.60 , and 14.45 for x = 0.35, 1.0, and 2.0,respectively. Predict the dependence of...
-
Draw a sketch of the fcc structure of C 60 . Show the positions of octahedral and tetrahedral holes and state the number of each. Assuming that C 60 has a radius of 5 , estimate the cubic cell...
-
A 230.0-g object on a spring oscillates left to right on a frictionless surface with a frequency of 2.00 Hz. Its position as a function of time is given by x = (8.00 cm) sin ( t.? (a) Sketch a graph...
-
You expect a share of Econ News.Com to sell for $65 a year from now. If you are willing to pay $60.38 for one share of the stock today, and you expect a dividend payment of $2.00, what rate of return...
-
Why is understanding the theory and application of Change Management practices so necessary to be a successful leader in today's global market? Why Is It especially Important for a Quality...
-
Choose a company you are familiar with, which uses out-of-home and/or Support media. 1. Find samples of at least three out-of-home/support media vehicles this company uses and briefly describe these...
-
3 months. Bill Dwyer aged 59 retired on 31 March 2023 and provided the following information. Gross Salary 62,300 Long service Leave 37100 Annual leave Tax withheld Wages and leave Gross...
-
1. What are the advantages and disadvantages of principal-protected equity-linked notes from the investor's (e.g. private banking client's) point of view? 2. If a client would like to invest...
-
Why is net income a good place to start when attempting to determine the cash flows from operating activities using the indirect method? Why is it not the same as the net cash flow from operating...
-
Anne is employed by Bradley Contracting Company. Bradley has a $1.3 million contract to build a small group of outbuildings in a national park. Anne alleges that Bradley Contracting has discriminated...
-
Calculate the pressure exerted by Ar for a molar volume of 1.31 L mol 1 at 426 K using the van der Waals equation of state. The van der Waals parameters a and b for Ar are 1.355 bar dm 6 mol 2 and...
-
A van der Waals gas undergoes an isothermal reversible expansion under conditions such that z > 1. Is the work done more or less than if the gas followed the ideal gas law?
-
When isopropylbenzene (cumene) is treated with NBS and irradiated with UV light, only one product is obtained. Propose a mechanism, and explain why only one product is formed. Br NBS hv
-
A 1 2 - year bond has a 9 percent annual coupon, a yield to maturity of 4 . 2 percent, and a face value of $ 1 , 0 0 0 . What is the price of the bond?
-
establish a step-by-step process to regularly review sources of information on taxation systems to ensure that payroll operations remain compliant
-
A chiropractic student receives a 1 0 - year PLUS Loan for $ 6 0 , 0 0 0 to complete the last 2 years of the program. If the interest rate of the loan is 5 . 4 5 % and the student begins repaying the...

Study smarter with the SolutionInn App


