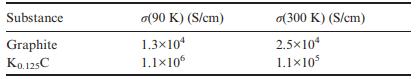
The table below lists the in-plane conductivity for graphite and K 0.125 C at two different temperatures.
Question:
The table below lists the in-plane conductivity for graphite and K0.125C at two different temperatures. Comment on these values.
Transcribed Image Text:
Substance Graphite K0.125C (90 K) (S/cm) 1.3x104 1.1106 o(300 K) (S/cm) 2.510 1.110
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The potassium intercalate has a higher conductivity than graphite at both temperatures This ca...View the full answer

Answered By

Pranav Makode
I am a bachelor students studying at professor ram meghe institute of technology and research. I have a great experience of being an expert. I have worked as an expert at helloexperts and solvelancer as a part time job. I have also worked as a doubt solver at ICAD SCHOOL OF LEARNING, which is in Amravati city. I have also worked as an Freelancer.
I have great experience of helping students, as described above. I can help any students in a most simple and understandable way. I will not give you have any chance for complaint. You will be greatfull to accept me as an expert.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
The table below lists body temperatures of seven subjects at 8 AM and at 12 AM (from Data Set 3 "Body Temperatures in Appendix B). The data are matched pairs because each pair of temperatures is...
-
The table below lists the U.S. median annual family income every 5 years during the period 1975-2015. It also contains several values for each of two simple indexes for median family income. a....
-
The table below lists measurements from eight different eruptions of the Old Faithful geyser in Yellowstone National Park. The data consist of the duration (sec) of the eruption, height (ft) of the...
-
At December 31, 2018, Landy Products has cash of $24,000, receivables of $18,000, and inventory of $80,000. The companys equipment totals $182,000. Landy owes accounts payable of $22,000 and...
-
A certain atom has three electrons (s, p, and d), in addition to filled shells, and is in a state with the greatest possible total mechanical moment for a given configuration. In the corresponding...
-
The Container Store is consistently profitable, enjoying a sterling reputation with customers and employees alike. With such proof, why would any major retailer not choose to institute similar core...
-
Calculate the times-interest-earned ratio for Evans \& Sons, Inc., for 2018 and 2019, and comment on the company's ability to pay its current interest payments. Did the company's ability to pay its...
-
The following information relates to the debt investments of Wildcat Company. 1. On February 1, the company purchased 10% bonds of Gibbons Co. having a par value of $300,000 at 100 plus accrued...
-
Explain the differences and similarities of information content theory in determining capital structure and determining dividend policy. What are the implications of each of these theories in...
-
The table below lists d spacings of the first four strong diffraction peaks observed in the diffraction patterns of graphite and two Rb intercalation compounds. Note that the graphite interlayer...
-
At high temperature, La 2 Mo 2 O 9 has a structure closely related to cubic -SnWO 4 , which contains WO 4 tetrahedra and Sn in a distorted six-coordinate environment. On cooling through 580 C, a 2a ...
-
Assume that Dorothy assigns you to help her with this forecasting problem. Write a report that recommends a course of action. Keep in mind that Marv must develop forecasts for the number of clients...
-
create a variable which equals to Scheduled Surprise, and then run a regression in which Y is Return, the first X variable is Surprise, and the second X variable is Scheduled*Surprise. The results...
-
Below is the monthly returns of Delta and IBM. Assume that the risk free (monthly) rate is 0.0015. Using the calculations (the mean for each stock is the expected return) plot (i) Return-Standard...
-
Cotrone Beverages makes energy drinks in three flavors: Original, Strawberry, and Orange. The company is currently operating at 75 percent of capacity. Worried about the company's performance, the...
-
Consider the monthly simple returns of CRSP decile 1, 2, 5, 9 and 10 portfolios based on the market capitalization of NYSE/AMEX/NASDAQ. The data span is from January 1961 to September 2011. For the...
-
Read the Essay regarding the Tesco UK retailer Groceries? ?and develop a structure of argument ? based on the information collected and analyzed.? Develop a position statement and use three...
-
The accounting records of The Picket Fence, Inc., reveal the following: Requirements 1. Compute cash flows from operating activities by the direct method. 2. Evaluate the operating cash flow of The...
-
An access route is being constructed across a field (Figure Q8). Apart from a relatively firm strip of ground alongside the field's longer side AB, the ground is generally marshy. The route can...
-
Explain why attractive interactions between molecules in gas make the pressure less than that predicted by the ideal gas equation of state.
-
Draw resonance structures for each of the following radicals: (a) (b) (c) (d)
-
Calculate the pressure exerted by benzene for a molar volume of 2.00 L at 595 K using the RedlichKwong equation of state: The RedlichKwong parameters a and b for benzene are 452.0 bar dm 6 mol 2 K...
-
$3,025,000 Pate, which choice shall you choose and why? received immediately. Suppose you 9. What's the rate of return you will have if you invest in an asset with the following expected cash flows...
-
The annual rainfall in a particular area of the country is normally distributed, with mean 100 cm and standard deviation 20 cm. A drought is said to occur after a year in which the annual rainfall...
-
Revenue $525.000 Sales Returns $13,900 Sales Discounts $12.400 Net Sales $498.700 Cost of Goods Sold $79.500 Gross Profit Gross Profit Margin % 84.0696

Study smarter with the SolutionInn App


