Use the MO diagram of oxygen to determine the oxygenoxygen bond order in the peroxide ion, O
Question:
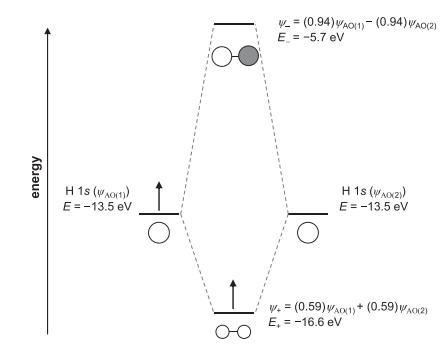
Use the MO diagram of oxygen to determine the oxygen–oxygen bond order in the peroxide ion, O22 . Will the O–O distance in peroxide be longer or shorter than in O2?
MO diagram
Transcribed Image Text:
energy H 1s ((1)) E = -13.5 eV Ho _= (0.94) WAO(1)- (0.94) WAO(2) E = -5.7 eV To H 1S (WA(Z)) E = -13.5 eV W+= (0.59) WAO(1) + (0.59) A0(2) E, = -16.6 eV
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Peroxide has two more electrons than O 2 Ther...View the full answer

Answered By

Ashish Bhalla
I have 12 years work experience as Professor for Accounting, Finance and Business related subjects also working as Online Tutor from last 8 years with highly decentralized organizations. I had obtained a B.Com, M.Com, MBA (Finance & Marketing). My research interest areas are Banking Problem & Investment Management. I am highly articulate and effective communicator with excellent team-building and interpersonal skills; work well with individuals at all levels.
4.80+
17+ Reviews
46+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
The oxygenoxygen bond in O2+ is 112 pm and in O2 is 121 pm. Explain why the bond length in O2+ is shorter than in O2. Would you expect the bond length in O2 to be longer or shorter than that in O2?...
-
Use the molecular orbital diagram in Fig. 17.5 to determine the bond order of the Br 2 + ion. Will the BrBr bond be longer or shorter than that in the Br 2 molecule? Figure 17.5. Energy 3s 20 1 1...
-
(a) Construct a Lewis structure for hydrogen peroxide, H2O2, in which each atom achieves an octet of electrons. (b) Do you expect the O-O bond in H2O2 to be longer or shorter than the bond in O2?
-
The input file for this assignment is Weekly_Gas_Average.txt. The file contains the average gas price for each week of the year. Write a program that reads the gas prices from the file into an...
-
Determine by direct integration the product of inertia of the given area with respect to the x and y axes.
-
Ellen wears eyeglasses with the prescription -1.0 D. a. What eye condition does Ellen have? b. What is her far point without the glasses?
-
12. ROLE REVERSAL: Write a multiple-choice question that raises the issue of an instrument's negotiability.
-
Reigle Electronics is a manufacturer of cell phones, a highly competitive business. Reigles Phones carry a price of $99, but competition forces the company to offer significant discounts and rebates....
-
00 Inventory Valuation under Abseron Costing and var sting At the end of the first year of operations, 4.400 units remained in the finished goods inventory. The unit manufacturing costs during the...
-
Construct an MO diagram for trigonal-planar BH3 by analogy with the MOs for trigonal-planar NH 3 in Figure 5.26. Use this diagram to determine the degeneracy and orbital character of the HOMO and the...
-
What are the values of the principal and orbital angular-momentum quantum numbers for each of the following orbitals? How many radial nodes and nodal planes does each orbital possess? (a) 4s orbital,...
-
Cineplex Inc. is the largest motion picture exhibitor in Canada and owns, leases, or has joint-venture interests in over 161 theatres with 1,630 screens. Exhibits 10-12A and B contain Notes 18 and 25...
-
An analysis of your favorite poem, short story, book, or essay from the course - You will not summarize the piece. Instead, you will write about its lasting impact on you and how it affected your...
-
Rough Draft of Researched Argument Assignment: Argue your own position on the topic you chose for this semester. Incorporate research from your annotated bibliography, include parts of your...
-
Maple Products Ltd produces hockey sticks. Data for the month of February for production of their top model, the "Canadian Pro D-Lux" is given below. The company uses JIT production and backflush...
-
Write- a paper, however, I need to plan outline of it first but I have no clue how to even start it. For some background information, the paper will be about a conspiracy theory that has evidence...
-
Question 1 (Essay Worth 10 points) (06.02 HC) Let= 11 12 Part A: Determine tane using the sum formula. Show all necessary work in the calculation. (5 points) Part B: Determine cos e using the...
-
What does risk taking have to do with the use of operating and financial leverage?
-
Dan and Diana file a joint return. Dan earned $31,000 during the year before losing his job. Diana received Social Security benefits of $5,000. a. Determine the taxable portion of the Social Security...
-
Using acetylene and methyl bromide as your only sources of carbon atoms, propose a synthesis for each of the following compounds: (a) (b) En Et Me En Et Me
-
Draw a mechanism and predict the major product for each reaction. a. b. c. d. e. f. ? 1) LAH H. 2) H20
-
Propose a molecular formula for a compound that exhibits the following peaks in its mass spectrum. a) (M) + at m/z = 72, relative height = 38.3% of base peak (M+1) + at m/z = 73, relative height =...
-
Using the balance sheet for STU Enterprises as of December 31, 2023, assess the companys financial leverage and stability. STU Enterprises Balance Sheet As of December 31, 2023 Assets Current Assets...
-
Jerry's survey asks the following question about income: What was your income in 2018 (in dollars)? 0-10,000 10,001-20,000 20,001-30,000 30,001-40,000 40,001-50,000 What is the level of measurement...
-
In chapter 19, what were the risks associated with the use of short-term forward contracts? Question 7 options: A) They would lock Kilgore into a more long-term price and could eliminate bigger...

Study smarter with the SolutionInn App


