Show that for a three-dimensional isotropic electron gas, (5.5.16) becomes (5.5.19) when f(E) e E/kB T,
Question:
Show that for a three-dimensional isotropic electron gas, (5.5.16) becomes (5.5.19) when f(E) ∝ e−E/kBT, that is, when the electrons have a Maxwell– Boltzmann distribution.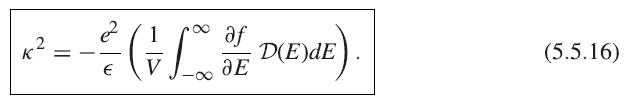

Transcribed Image Text:
K 2 = af - ²/² ( = // * / * PXEME.) D(E)dE € V JE (5.5.16)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
When the distribution of OE tive in Eq 584 is simply l...View the full answer

Answered By

Anurag Agrawal
I am a highly enthusiastic person who likes to explain concepts in simplified language. Be it in my job role as a manager of 4 people or when I used to take classes for specially able kids at our university. I did this continuously for 3 years and my god, that was so fulfilling. Sometimes I've skipped my own classes just to teach these kids and help them get their fair share of opportunities, which they would have missed out on. This was the key driver for me during that time. But since I've joined my job I wasn't able to make time for my passion of teaching due to hectic schedules. But now I've made a commitment to teach for at least an hour a day.
I am highly proficient in school level math and science and reasonably good for college level. In addition to this I am especially interested in courses related to finance and economics. In quest to learn I recently gave the CFA level 1 in Dec 19, hopefully I'll clear it. Finger's crossed :)
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Three Dimensional Image The longitudinal magnification is defined as m' = ds'/ds. It relates the longitudinal dimension of a small object to the longitudinal dimension of its image. (a) Show that for...
-
Show that for a monatomic model solid the Young modulus E is given approximately by E = 1/r0(d2U/dr2)x = x0 where U is the interaction potential energy of a pair of atoms distance r apart and r0 is...
-
How does the range of hFE (Fig. 3.23j, normalized from hFE = 100) compare with the range of hfe (Fig. 3.23f) for the range of IC from 0.1 to 10 mA?
-
Bench Inc., an equipment manufacturer, leased a machine to Unicycle Company on January 1, 2014. The lease is for a 6-year period and requires equal annual payments of $41,747 at the beginning of each...
-
please show work. A project requires a $2,880,000 initial investment for new machinery with a five-year life and a salvage value of $316,000. The project is expected to yield annual income of...
-
Identify which of the following numbers are irrational: \(\sqrt{441}, 4.33, \sqrt{70}, 5+9 \pi\)
-
Marvel Parts, Inc., manufactures auto accessories. One of the companys products is a set of seat covers that can be adjusted to fit nearly any small car. The company has a standard cost system in use...
-
Mercedes and Alejandro San Martin It is January 2021. Mercedes and Alejandro have lived in Toronto, Ontario for the last 25 years since immigrating to Canada from South America in 1996. Married in ...
-
In a cubic crystal, a triply degenerate band with p-symmetry has the following deformation Hamiltonian, known as the PikusBir Hamiltonian (derived in Section 6.11.2): operating on the basis of the...
-
Estimate the melting temperature of a solid with lattice constant 6 , optical phonon energy 30 meV, and effective mass of the unit cell given by the mass of a silicon atom. Does your answer seem...
-
In Problems 714, find the value of each determinant. -4 -4 2 -5 3
-
What was the basis of race classification in ancient societies?
-
A system modelled as x ( t ) = A x ( t ) x ( t ) = A x ( t ) generates state response for initial vector Missing \end{array} and for Missing \end{array} . Find the system matrix A and state...
-
What are some of the differences between the peripheral and semiperipheral countries in Latin America, Africa, and the Caribbean?
-
How do property rights in agricultural states differ from those in forager and tribal societies?
-
What do anthropologists mean by the term globalization?
-
What is meant by the assignee takes subject to the equities?
-
What did Lennox gain by integrating their WMS, TMS, and labor management systems?
-
You have containers of pure O 2 and N 2 at 298 K and 1atm pressure. Calculate G mixing relative to the unmixed gases of a. A mixture of 10.mol of O 2 and 10.mol of N 2 b. A mixture of 10.mol of O 2...
-
You get a PT phase diagram by projecting a PVT phase diagram on the PT plane.
-
The mechanism of the following transformation involves a carbocation intermediate that rearranges in a way that we have not yet seen. Rather than occurring via a methyl shift or a hydride shift, a...
-
The force developed in hydraulic systems is high due to _ _ _ _ a ) high pressure b ) more oil c ) less pressure d ) less oil
-
Much of the rapid growth since the late nineteenth century has come from harnessing the power of competitive markets to _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ . This market orientation typically reaches...
-
Question 1 The feasibility study captures a view of what has gone well and what needs to be improved in order to successfully complete a project. True False Moving to another question will save this...

Study smarter with the SolutionInn App


