Show that not only xx , but also xx vanishes in the limit
Question:
Show that not only σxx , but also ρxx vanishes in the limit τ → ∞, where ρxx is the diagonal resistivity defined by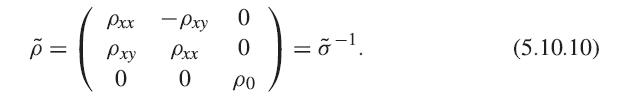
Transcribed Image Text:
õ = Pxx Pxy 0 - Pxy Pxx 0 0 :) = 0 | =õ–¹ po (5.10.10)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The full matrix is 1 wer2 1 wer2wet 0 1 wer2wet 1 w...View the full answer

Answered By

Asd fgh
sadasmdna,smdna,smdna,msdn,masdn,masnd,masnd,m asd.as,dmas,dma.,sd as.dmas.,dma.,s ma.,sdm.,as mda.,smd.,asmd.,asmd.,asmd.,asm
5.00+
1+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Using Table 1.6, write a structural formula for each of the following: a. An alcohol, C3H8O b. An ether, C4H10O c. An aldehyde, C3H6O d. A ketone, C3H6O e. A carboxylic acid, C3H6O2 f. An ester,...
-
Chemical Financial Corporation signs an agreement on January 1, 2014, to lease equipment to Chells, Inc. The following information relates to this agreement. 1. The term of the noncancelable lease is...
-
Which preventive, detective, and/or corrective controls would best mitigate the following threats? a. An employees laptop was stolen at the airport. The laptop contained personally identifying...
-
\(\sqrt{45}\) Simplify the square root by expressing it in lowest terms.
-
The following control procedures are used in Kelton Company for over-the-counter cash receipts. 1. Each store manager is responsible for interviewing applicants for cashier jobs. They are hired if...
-
Can anyone solve these questions please? Thank you P 4-13 (similar to) Question Helpv Q You wanl to endow a scholarship that will pay $11,000 per year forever, marling one year from now, lithe...
-
For a typical resistance R = 100 in a resistor of length l = 2 mm and cross-sectional area A = 1 mm 2 , where R = l/A , calculate the average scattering time for an electron, for a free carrier...
-
If the Rayleigh scattering of light in the atmosphere is known to be dominated by scattering from a particulate with radius 50 nm and index of refraction n = 2, estimate the particulate density if...
-
In each of the following instances, determine whether supply is elastic, inelastic, or unitelastic. a. If price increases by 10 percent and quantity supplied increases by 15 percent, supply is...
-
What does the anatomy of the Paleocene fossils suggest about what selective pressures may have led to the origin of Primates and how Eocene primates differed from the Paleocene groups?
-
What can we learn from the ethnic group adaptations in U.S. society?
-
What species might be the earliest hominins, and what does their anatomy and environment tell us about the selective pressures that might have facilitated their evolution?
-
Keynesianism has been subject to much debate over the years. To what extent is the idea that demand management in a period of economic stagnation a recipe for stimulating economic growth once again?...
-
What role did kinship and descent play in the social structure of a chiefdom?
-
A mistake may result in a contract being declared void or voidable. What difference does it make if a contract is merely voidable?
-
Calculate I, , and a for a 0.0175 m solution of Na 3 PO 4 at 298 K. Assume complete dissociation. How confident are you that your calculated results will agree with experimental results?
-
Draw the mechanism for each of the following transformations: a. b. c. Br HBr
-
Using the result of Equation (3.8), (P/T) V = / , express as a function of and V m for an ideal gas, and as a function of b, , and V m for a van der Waals gas.
-
Is the following statement correct? If not, rewrite it so that it is correct. If H o R for a chemical reaction does not change appreciably with temperature, the heat capacities for reactants and...
-
Last year, a Pacific Island nation imported over $3.2 billion of goods and reported a trade surplus. This would suggest that the nation had OA. money spent by tourists and the value of its exports...
-
Why did the accusations of witchcraft in Salem suddenly snowball in 1692? O a. All of the accused were children, and Puritans were determined to force their young to accept their religious traditions...
-
Table 1 shows the data of pressure, P, as a function of temperature, T, for the experiment of determining the absolute zero temperature. According to Charles's law for an ideal gas, at constant...

Study smarter with the SolutionInn App


