Attempted CrO 3 oxidation of 1,4-butanediol to butanedioic acid results in signifi cant yields of g-butyrolactone. Explain
Question:
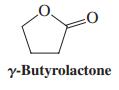
Attempted CrO3 oxidation of 1,4-butanediol to butanedioic acid results in signifi cant yields of “g-butyrolactone.” Explain mechanistically.
Transcribed Image Text:
:0 y-Butyrolactone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
HO CH HOCH CH 0CH CH CH H CO...View the full answer

Answered By

DIPAK SHETE
Hello guys, I dipak master in Chemistry. I have three years teaching experience in science and six months experience in online tutor (Q&A Expert). I able to solve any doubt in chemical sciences and mathematics also.
I have solved several problems of peoples regarding chemical sciences. I totally believe on knowledge increases
By sharing with others. And I always search someone with I can share my knowledge.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Explain the outcome of the following transformations mechanistically. (a) (b) (c) CHCH OH HSCH CH2BrNaOH DMF BrCH-CH,CH-CH,CHBr + NaOH- Excess BrCH CH CH CH CH,Br+ NH, CHCHO Excess
-
When 1,4- and 1,5-dicarboxylic acids, such as butanedioic (succinic) acid (Section 19-8), are treated with SOCl2 or PBr3 in attempted preparations of the diacyl halides, the corresponding cyclic...
-
Oxidation of an aldehyde yields a carboxylic acid: Draw the structures for the products of the following oxidation reactions. a. b. c. [ox] propanal 2,3-dimethylpentanal ox] 3-ethylbenzaldehyde>
-
If you were in charge of B&D, indicate the possible decisions you might take as regards the four Ps and the points you would want to keep in mind as you go about the task of marketing for...
-
Write down three industries in which you suspect that strategic trade policy is a tool that governments could potentially use, and three in which you suspect that it is not. Explain your reasoning in...
-
Doggie Nuggets Inc. (DNI) sells large bags of dog food to warehouse clubs. DNI uses an automatic filling process to fill the bags. Weights of the filled bags are approximately normally distributed...
-
Most costs are locked in at which stage in the production cycle? a. product design b. production planning c. production operations d. cost accounting
-
Aldrich and Co. sold goods to Donovan on credit. The amount owed grew steadily, and finally Aldrich refused to sell any more to Donovan unless Donovan signed a promissory note for the amount due....
-
Response of Delays to Changing Delay Times To develop your understanding of how the different types of delays respond to variations in the delay time, answer the following questions. 1. Consider a...
-
Why might an organization seek the assurance of a CPA about information presented in the organizations corporate sustainability report?
-
How would you expect the acidity of acetamide to compare with that of acetic acid? With that of acetone? Which protons in acetamide are the most acidic? Where would you expect acetamide to be...
-
Following the general mechanistic scheme, write detailed mechanisms for each of the following substitution reactions. (These transformations are part of Chapter 20, but try to solve the problem...
-
Douglas Davis, controller for Marston, Inc., prepared the following budget for manufacturing costs at two different levels of activity for 2010: During 2010, Marston worked a total of 80,000 direct...
-
Dodge, CPA, is examining the financial statements of a manufacturing company with a significant amount of trade accounts receivable. Dodge is satisfied that the accounts are properly summarized and...
-
The viscosity of liquids and the viscosity of gases \( \qquad \) (a) Increases, increases (b) Increases, decreases (c) Decreases, increases (d) Decreases, decreases (e) Decreases, remains the same
-
What is Mach tuck? What is the adverse "stick force"?
-
What is the Mach buffet?
-
Which is of the highest value? (a) 1 bar (b) \(10^{5} \mathrm{~N} / \mathrm{m}^{2}\) (c) \(1 \mathrm{~atm}\) (d) \(100 \mathrm{kPa}\) (e) None of these
-
Explain the general stages of team development.
-
A supermarket chain is interested in exploring the relationship between the sales of its store-brand canned vegetables (y), the amount spent on promotion of the vegetables in local newspapers (x1)...
-
Name these compounds: CH3 a) CHCHCHCHCHCH, b) NHCH, NH
-
Draw structures for these compounds: (a) Diethyl ammonium bromide (b) N-Methyl-3-(1-methylpropyl)-2-octanamine
-
Name thesecompounds: CH3 CH3 T a) CHCH,CH_CHCHCH,CH, CHCH3 b) CHCH=CHCHCHCH3 CI c) CH,CH,CHC=CH d) e) f) g) OH CH3 H
-
Provide an example of how you used the observation tool to record meaningful and accurate information. Attach proof of your recorded observations to your portfolio. Make sure you document your...
-
you buy 3 0 0 0 0 shares of rangers company preferred stock at $ 4 4 per share. every quarter you get a $ 1 . 2 5 preferred dividend for each share of stock. exactly one year later you sell the...
-
Suppose a project has the following cash flows: Year 0 : - 2 5 million dollars Year 1 : 8 million dollars Year 2 : 1 0 million dollars Year 3 : 1 2 million dollars Year 4 : 1 4 million dollars What...

Study smarter with the SolutionInn App


