Farnesol is a molecule that makes flowers smell good (lilacs, for instance). Treatment with hot concentrated H
Question:
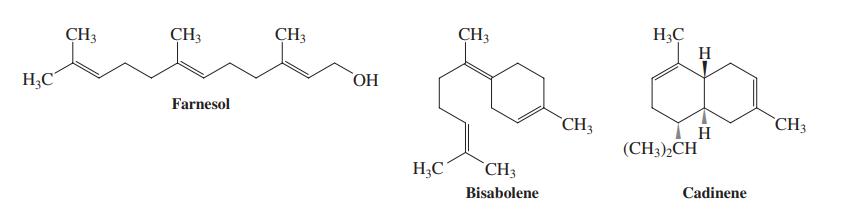
Farnesol is a molecule that makes flowers smell good (lilacs, for instance). Treatment with hot concentrated H2SO4 converts farnesol fi rst into bisabolene and finally into cadinene, a compound of the essential oils of junipers and cedars. Propose detailed mechanisms for these conversions.
Transcribed Image Text:
H3Ç H CH3 CH3 ÇH3 CH3 HO, HC Farnesol CH3 `CH3 H (CH3),CH H;C CH3 Bisabolene Cadinene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
The wildlife we love and their habitat will be dest...View the full answer

Answered By

Dudhat Vaidehi
I tutored mostly elementary school students privately after school and during the summer. We met in their homes or at the public library. I charged an hourly fee, and I provided any necessary materials.
Having taught in special education in two local schools for many years meant that I had contact with a lot of parents of special needs students. I never had to advertise — word of mouth was how most folks knew of me. At one point I did have a website, but didn't utilize it much. I stayed very busy, especially in the summers, and always had a full schedule. I typically met with each student's teacher in order to get an idea of what the focus of my instruction/remediation should be. Becoming familiar with the student's learning style(s) was also very helpful. Often parents would share records and test results with me. After each tutoring session, I documented the student’s progress and gave parents written updates, as well as phone calls or emails as needed.
While my students and I certainly utilized technology and the internet often during our sessions, I never tutored online or for any tutoring company, so am not familiar with the curriculums or methods used in those settings.
Tutoring one on one was very enjoyable and rewarding. My students and I had fun, and grew quite fond of one another. The extra income was a bonus. I had to retire from tutoring due to a physically handicapping disease, and miss my students very much.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Propose mechanisms to account for the observed products in the following reactions. In some cases more products are formed, but you only need to account for the ones shown here. (a) (b) (c) (d) Br hv...
-
Compound A is an optically active alcohol. Treatment with chromic acid converts A into a ketone, B. In a separate reaction, A is treated with PBr3, converting A into compound C. Compound C is...
-
The concentration of concentrated H2SO4 is about 18 M. Assuming only one H+ comes off the H2SO4 molecule, what is its pH and pOH? What would the pH and pOH be if the second H+ were also ionized?
-
The Wall Street Journal Corporate Perceptions Study 2011 surveyed readers and asked how each rated the quality of management and the reputation of the company for over 250 worldwide corporations....
-
List the reasons for the creation of the Project Management Institute.
-
How much will you have in an account after 25 years if you put $1500 per month in the account earns 6% compounded monthly? Answer = $ b) How much will you have after 25 years if you put $1500 per...
-
List specific bylaws clauses that are bound to change as the organization evolves. How might these issues be handled in a start - up board and then in a more mature board?
-
Refer to Exercise 6-1. Calculate the amount of the noncontrolling interest to be deducted from consolidated income in arriving at 2011 controlling interest in consolidated net income.
-
Think of an innovation, such as some form of technology- or problem-based learning. How could LoU be used as a tool to monitor its implementation? Explain how you see this used in an actual school...
-
Great Ape Glassworks manufactures glass used for the screens on smartphones. It has two producing departments, P1 and P2, and only one service department, S1. The company is able to track overhead to...
-
In a published synthetic procedure, acetone is treated with ethenyl (vinyl) magnesium bromide, and the reaction mixture is then neutralized with strong aqueous acid. The product exhibits the 1 H NMR...
-
The ratio of 1,2- to 1,4-addition of Br 2 to 1,3-butadiene is temperature dependent. Identify the kinetic and thermodynamic products, and explain your choices.
-
The following numbers were calculated from the financial statements for a firm for 2012 and 2011: Calculate core return of net operating assets (core RNOA) and show how much of its change from 2011...
-
A CPA is permitted to disclose confidential client information without the con- sent of the client to: I: Another CPA who has purchased the CPA's tax practice. II. Another CPA firm if the information...
-
The Sarbanes-Oxley Act prohibits public accounting firms from providing cer- tain services to audit clients that are public companies. Which of the following services is not prohibited? a. Internal...
-
What are the general characteristics of diode clamper circuits?
-
Which of the following is not a major threat to an auditor's independence? a. Audit partner's compensation based on obtaining and retaining clients b. Becoming too friendly with the client's...
-
Which of the following statements best explains why the CPA profession has found it essential to promulgate ethical standards and to establish means for en- suring their observance? a. Vigorous...
-
What are the Miller indices for the plane shown in the following cubic unit cell? (A) (201) (B) (1 ( 1 / 2) (C) (10 1/2) (D) (102) -/2
-
Do public and private companies follow the same set of accounting rules? Explain.
-
Calculate d for a hydrogenic atom with atomic number Z.
-
NMR spectroscopy may be used to determine the equilibrium constant for dissociation of a complex between a small molecule, such as an enzyme inhibitor I, and a protein, such as an enzyme E: El E + I...
-
Use concepts of statistical thermodynamics to describe the molecular features that lead to the equations of state of perfect and real gases.
-
1. Estimate Li's net worth at age 67 if his net worth grows at 5% per year, his stock fund grows at 10% per year, and his bond fund grows at 6% per year. 1. 2. Li is amazed that he will be able to...
-
Assume a parent company acquired 75% of the common stock of a subsidiary a few years ago. On the acquisition date, the fair value of the controlling interest and noncontrolling interest equaled the...
-
Explain the concept capital account of the balance of payments.

Study smarter with the SolutionInn App


