Give the likely products of each of the following reactions. All are carried out in the pre
Question:
Give the likely products of each of the following reactions. All are carried out in the pre sence of a Pd catalyst, a ligand for the metal such as a phosphine, and heat.
(a)
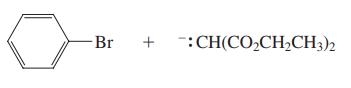
(b)
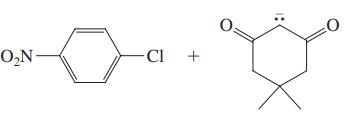
Transcribed Image Text:
Br + :CH(CO2CH2CH3)2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
a b O 0 ...View the full answer

Answered By

KULDEEP MITTAL
CSIR UGC NET with AIR 46 and AIR 19
GATE 2020 with AIR 5
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the likely products of each of the following reactions. Each one is carried out in the presence of a Pd catalyst, a phosphine, and heat. (a) (b) (c) (d) F;C- Cl 'N' H
-
Give the likely products for each of the following aldol addition reactions. NaOH, H,O () 2 -CH,CHO NaOH, H,O (b) - + (H3)CHO NaOH, H,O CH3 NaOH, H,O () H3C (d) CH3
-
For each of the following reactions, give the substitution products, assuming that all the reactions are carried out under SN2 conditions; if the products can exist as stereoisomers, show what...
-
Why is globalization of industries a common factor today?
-
The world market for large passenger jet airplanes is an oligopoly dominated by two firms: Boeing in the United States and Airbus in Europe. a. Explain why the market equilibrium might involve either...
-
Solve each equation or inequality. |5 0.5x| = 4
-
When reviewing an organizational chart, what represents the formal line of authority and responsibility within the organization? A. Specialization of labor. B. Chain of command. C. Span of control....
-
Manufacturing cost data for Orlando Company, which uses a job order cost system, are presented below. Indicate the missing amount for each letter. Assume that in all cases manufacturing overhead is...
-
can having healthy diet prevent diseases like cancer? what are the limitation to the mobile applications out there that are there to help prevent diseases?
-
Logan B Taylor is a widow whose wife, Sara died on June 6, 2018. He lives at 4680 Dogwood Lane, Bakersfield, CA 93301. He is employed as a paralegal by a local law firm. During 2020, he had the...
-
Write out, in full detail, the mechanism of the Michael addition of malonic ester to 3-buten-2-one in the presence of ethoxide ion. Be sure to indicate all steps that are reversible. Does the overall...
-
Based on the mechanism presented for the Pd-catalyzed reaction of a halobenzene with hydroxide ion, write out a reasonable mechanism for the Pd-catalyzed reaction of Problem 38(a). Problem 38(a) (a)...
-
Three SSSF flows are mixed in an adiabatic chamber at 150 kPa. Flow one is 2 kg/s of O2 at 340 K, flow two is 4 kg/s of N2 at 280 K and flow three is 3 kg/s of CO2 at 310 K. All flows are at 150 kPa...
-
What is an intelligent agent? Identify two major ways in which intelligent agents might be utilized to enhance the efficiency of an audit engagement.
-
How should you approach conducting a site survey?
-
How do concurrent audit techniques differ from the more static audit techniques, such as test data or GAS? What audit objectives are accomplished by the concurrent audit techniques that might not be...
-
Which of the following is not an auditing procedure that is commonly used in performing tests of controls? a. Inquiry. c. Comparison. b. Observation. d. Inspection.
-
An auditor decides to use IDEA to test the details of a material account balance. How can the auditor be sure that he or she has captured the detail file that supports the general ledger account...
-
Understand how historical forces influence the practice of management ?
-
Find the market equilibrium point for the following demand and supply functions. Demand: 2p = - q + 56 Supply: 3p - q = 34
-
Why do you suppose no one has ever been able to make cyclopentyne as a stable molecule? Cyclopentyne
-
What is wrong with following sentences? The bonding molecular orbital in ethylene results from sideways overlap of two p atomic orbitals.
-
Allene, H2C = C = CH2, is somewhat unusual in that it has two adjacent double bonds. Draw a picture showing the orbitals involved in the and bonds of allene. Is the central atom sp2- or...
-
Although appealing to more refined tastes, art as a collectible has not always performed so profitably. Assume that in 2015, an auction house sold a statute at auction for a price of $10,584,500....
-
Johannesburg - Chile's CFR Pharmaceuticals said on Friday it would drop its $1.2bn bid for drugmaker Adcock Ingram [JSE:AIP] after being thwarted by Adcock's top shareholder. "Shareholders are...
-
Wilma is considering opening a widget factory. The unlevered cost of equity for making widgets is 0.13. This factory would cost $11 million to set up, and would produce EBIT of $3 million per year...

Study smarter with the SolutionInn App


