Give the product of each of the following reactions. (a) (b) (c) (d) (e) CH 1. ,
Question:
Give the product of each of the following reactions.
(a)
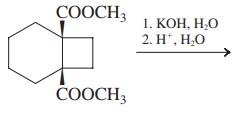
(b)
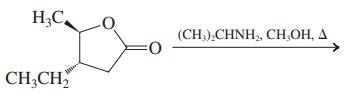
(c)
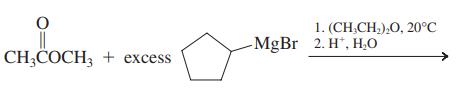
(d)
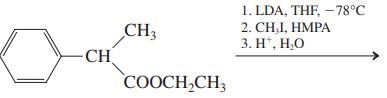
(e)
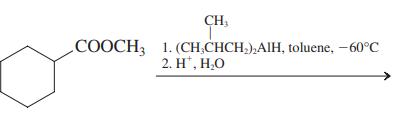
Transcribed Image Text:
СООCH 1. КОН, Н.О 2. H', Н.О СООCH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
a...View the full answer

Answered By

MANDWEEP BHUMIJ
I did my masters in Tezpur University with specialization in Organic chemistry. I cleared CSIR NET-JRF with all India ranking 74. I am currently working as Junior Research Fellowship in Central drug research institute Lucknow. I have some private tutoring experiences in multiple subjects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the product of each of the following reactions: (a) (b) (c) (d) CH3 CHCH3 Na2r207.H CH2CH3 Na,Cr0H CH3 1. NBS/A/peroxide 2. CH30 CH3 1. NBS/A /peroxide 3. H2/Ni
-
Give the product of each of the following reactions: a. b. c. d. OHH, . CHCHCH,N12 catalytic Zo outo 0 auda CH3CCI + 2 1. HCI, NaNO2 22. H20, Cu20, Cu(NO32 catalytic
-
Give the product of each of the following reactions: a. b. c. d. CH CH3 A CH2CH3 CH2CH 2CH3 -A CH2CH3 h CH2CH3 CH2CH3 CH2CHs
-
Your restaurant has five menu choices for lunch. How many ways can you order them on your menu? A 0.0749 B 0.0747 120
-
Consider the following thought experiment regarding foreign direct investment and specific factors. (a) In the pure specific-factors model in the text, suppose that a wave of foreign direct...
-
The bar graph shows your chances of surviving to various ages once you reach 60. The functions model the chance, as a percent, that a 60-year-old will survive to age x. Use this information to solve...
-
Information from Razure Adventures Companys financial statements for the current year are provided in the Working Papers. Work independently to complete the following problem. 1. Calculate the...
-
Suppose A and B live in an exchange economy with two goods x1 and x2. A owns 50 of both goods, and B own 250 of both goods. A's tastes are captured by the utility function uA(x1,x2) = x2 + 50lnx1 and...
-
Thetablebelowshowsfluctuationsinthenumberofconveniencestoresnationwidefrom2008to2011basedonaquantitativesurveyofconveniencestores. Trytodrawagraph.Whatinterpretationscanyoudrawfromthegraph?...
-
1. Using the spreadsheet model from Case 2.1 as a starting point, use Solver to find the optimal set of projects to approve. The solution should maximize the total NPV from the approved projects, and...
-
Reaction review. Suggest reagents to convert each of the following starting materials into the indicated product: (a) hexanoyl chloride into acetic hexanoic anhydride; (b) methyl hexanoate into...
-
For each of the naturally occurring lactones below, draw the structure of the compound that would result from hydrolysis using aqueous base. (a) (b) (c) Sedanenolide, major contributor to the flavor...
-
Zachary Wall is thinking about expanding the product offerings in his company's automotive supply store. He would like to include tires. In deciding which product lines to carry, Zachary knows that...
-
What are the limits on the efficiency of a screw jack to be of the self-locking type?
-
Fraudulent financial reporting has been described as a function of three requirements. Describe these requirements (not the primary or secondary indicators of each requirement).
-
What matters should an auditor consider when planning an audit?
-
Explain how stewardship or agency theory helps to explain the demand for audits.
-
Which of the following statements best describes an auditor's responsibility to detect errors and fraud? a. The auditor should study and evaluate the client's internal control, and design the audit...
-
Discuss how women's style of leading is typically different from men's.
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
Identify these pairs of compounds as identical, structural isomers, enantiomers, or diastereomers: CH3 CH3 H3 CH Br - - - - -Cl - b) Br- a) - - CI - - CH3 CH CH3 CH3 CH3 CH3 CH CI Br - - CH3...
-
(a) A solution of 0.2g/mL of a compound in a 1 dm cell rotates plane-polarized light + 13.3 o at the sodium D line. What is the specific rotation of this compound? (b) What is the rotation caused by...
-
Describe how this amine could be resolved by using this carboxylic acid? CH3 CNH, H HC H CI C COH
-
I have trouble importing the tpmon.dat in R. How to import this file in R file nametpmon.dat path"C:\Users\Lenovo\Downloads\tpmon.dat"
-
Based on the answers to the previous two questions, how does the proposed agency s marketing promotions meet community expectations?
-
Seth sold a plot of land with a basis of $24,000 on January 1, 2023, to Natalie for $56,000. Natalie agreed to pay $8,000 per year plus interest for years 2023 through 2029. What is Seth's taxable...

Study smarter with the SolutionInn App


