How would you expect the following classes of compounds to compare with simple primary amines as bases
Question:
How would you expect the following classes of compounds to compare with simple primary amines as bases and acids?
(a) Carboxylic amides; for example, CH3CONH2
(b) Imides; for example, CH3CONHCOCH3
(c) Enamines; for example, CH2PCHN(CH3)2
(d) 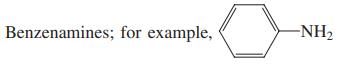
Transcribed Image Text:
Benzenamines; for example, -NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
A Due to resonance this molecule is less basic than prima...View the full answer

Answered By

Archana Prasad
Hi! My name is Archana Prasad. I did my graduation in Biotechnology in year 2011 from Patna University. After that I completed my Master degree from Patna Science College, Patna University in BIOCHEMISTRY in year 2013. During the session I participated in various seminars and internships at reputed platform such as Sudha Diary, Patna and perform well there.
As per my tutoring experience is concern I have expertise of 4 years at reputed coaching institutions in Patna, Bihar. I have also experience of solving questions on various online platform. I have worked with PhotoStudy as well as Studypool.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Would you expect the following carboxylic acids to be more acidic or less acidic than benzoic acid? Explain. (Reddish brown = Br) (a) (b) (d) (c)
-
(a)Construct a hybrid orbital picture for the hydronium ion (H3O+) using oxygen sp3 hybrid orbitals. (b) How would you expect the H-O-H bond angles in hydronium ion to compare with those in water...
-
The following classes of organic compounds are Lewis bases. Write an equation that shows how each class might react with H+. a. Amine, R3N: b. c. ether, ROR ketone, R2C O
-
Refer to the bolt strength problem 17.47. Assume = 6,050 and 5 100. Use the following 24 individual bolt strength observations to answer the questions posed. (a) Prepare a histogram and/or normal...
-
A country with a fixed exchange rate has achieved external balance. Government spending then increases in an effort to reduce unemployment. What is the effect of this policy change on the country's...
-
1.Identify, if a forward contract is properly priced based on put-call-forward parity. The option exercise price is $90, the risk-free rate is 5 percent, the options and the forward contract expire...
-
Situational and Contingency theory: A. Embodies the idea that the leader does the right thing based on the situation. B. Involves assessing the nature of the task and the followers motivation to...
-
Following are financial statement information for Welmark Corporation as of Year 2 and Year 3. Required: Using the residual income model, prepare a valuation of the common stock of Welmark...
-
How can non-profit organization diversify their funding to ensure revenue growth, reduce financial risks and expand capacity? Please profit a reference.
-
As a financial analyst, you have been brought in to help various organizations or individuals to make decisions on possible financial investments that have been presented to them. Solve each of these...
-
In which direction would you expect each of the following equilibria to lie? (a) NH 3 + - OH NH 2 - + H 2 O (b) CH 3 NH 2 + H 2 O CH 3 NH 3 + + - OH (c) CH 3 NH 2 + (CH 3 ) 3 NH + CH 3 NH 3 + +...
-
Several functional groups containing nitrogen are considerably stronger bases than are ordinary amines. One is the amidine group found in DBN and DBU, both of which are widely used as bases in a...
-
No financial decision-maker should ever rely solely on the financial information reported in the annual report to make decisions. It is important to keep abreast of financial news. This activity...
-
How have auditors responded to the increasingly litigious environment?
-
Given two vector spaces $V_{1}$ and $V_{2}$, prove that the dimension of their direct sum is $\operatorname{dim}\left(V_{1} \oplus V_{2} ight)=\operatorname{dim} V_{1}+\operatorname{dim} V_{2}$.
-
Describe the reforms being addressed by the Coalition to Eliminate Abusive Securities Suits (CEASS).
-
In an effective internal control system, the following functions are separated: (1) authorizing a transaction, (2) recording a transaction, (3) maintaining custody of assets that result from a...
-
In the Ultramares case the plaintiff's charge of negligence a. Was disallowed because the plaintiff was not a foreseen party. b. Was disallowed because the plaintiff had a privity relationship with...
-
Define organizational change and explain the forces driving innovation and change in today's organizations.
-
A test car is driven a fixed distance of n miles along a straight highway. (Here n Z+.) The car travels at one mile per hour for the first mile, two miles per hour for the second mile, four miles...
-
Show the products of these reactions: Br DMSO + CH,CO, a) CI acetone + CH,CH,CO, b) CO DMF c) CH,CH CH,CH,Br +
-
Show all the steps in the mechanism for the reactions of 2-methyl-2-butanol with HCl in water.
-
Suggest reagents that could be used to prepare these alkyl halides from alcohols: CH3 CH, b) CH,CI ) C,r Ph CH;CH2 Br CI Br ) d)
-
A family bought a car for 40,000. They made a down payment of 8,000. They take out a 5-year auto loan for $32,000 at an annual interest rate of 4%. Find the total payment when the loan is amortized...
-
Henry and Jane, MFJ taxpayer (taxable income is $305,000 before considering any of the items below), sold the following assets during the year: Asset Sale Price Tax Basis Gain or Loss Holding Period...
-
an investor buys a 7% semi-annual payment bond with three years to maturity. the bond has a yield to maturity to 8.5% and is currently priced at 96.100 per 100 of par. calculate the bonds macaulay...

Study smarter with the SolutionInn App


