Identify by name (either IUPAC or common) as many of the heterocyclic rings contained in the structures
Question:
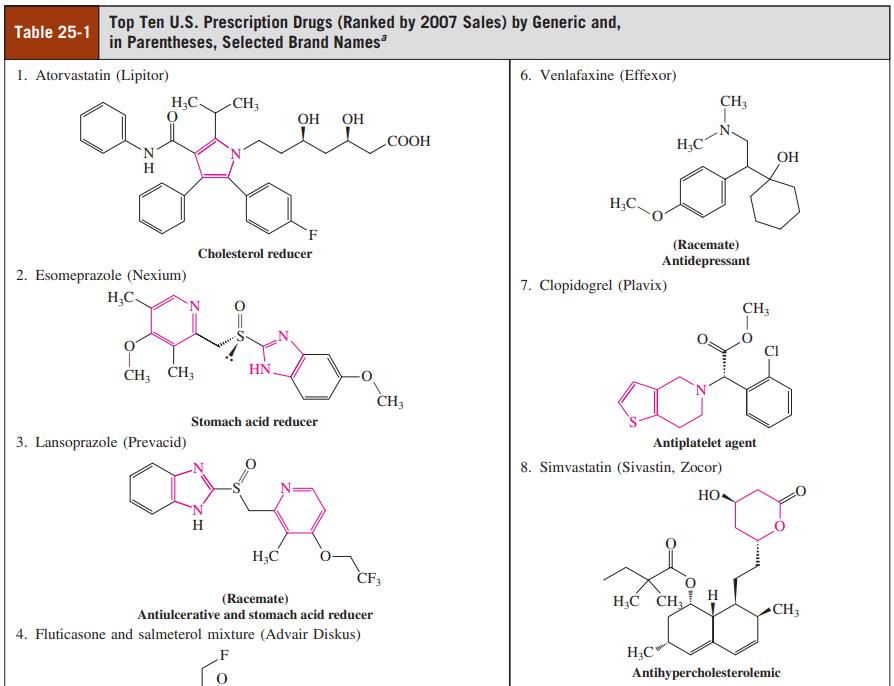
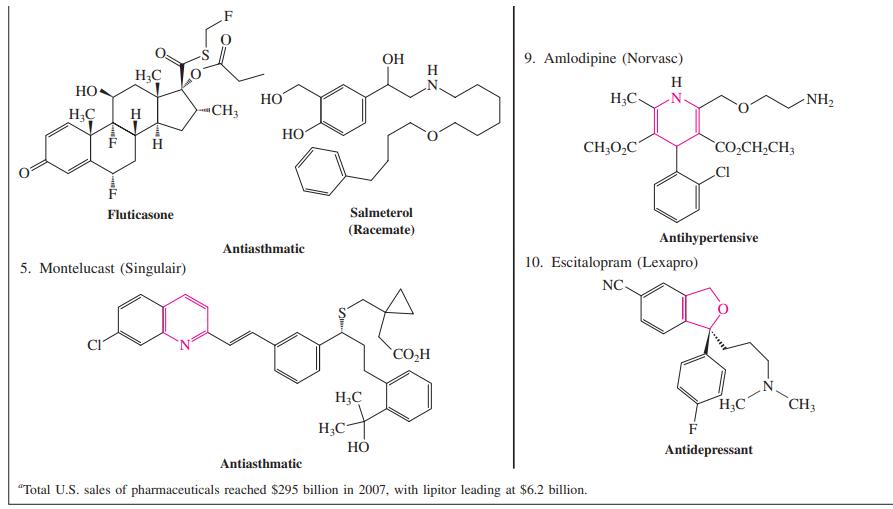
Identify by name (either IUPAC or common) as many of the heterocyclic rings contained in the structures shown in Table 25-1 as you can.
Transcribed Image Text:
Top Ten U.S. Prescription Drugs (Ranked by 2007 Sales) by Generic and, in Parentheses, Selected Brand Names Table 25-1 1. Atorvastatin (Lipitor) 6. Venlafaxine (Effexor) H3C- -CH3 CH3 Он ОН COOH H;C N. OH H;C `F (Racemate) Antidepressant Cholesterol reducer 2. Esomeprazole (Nexium) 7. Clopidogrel (Plavix) H,C- 'N. CH; CI HN. CH; CH; CH; Stomach acid reducer 3. Lansoprazole (Prevacid) Antiplatelet agent 8. Simvastatin (Sivastin, Zocor) HO H H;C CF3 (Racemate) H;C CH H Antiulcerative and stomach acid reducer CH3 4. Fluticasone and salmeterol mixture (Advair Diskus) H;C' Antihypercholesterolemic
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
1 Pyrrole 2 Pyridine and benzimidazole S Pyridi...View the full answer

Answered By

Sahil Singh
Hi! This is Sahil Singh. I've a tutoring experience of 4 years. I look to create a sense of self-belief in students while solving questions. I work on student's problems quite diligently and solve the problems in easily understandable form. That's why I put more emphasis on building stronger basics and fundamentals of the concepts. So, don't hesitate in reaching out to me by contacting me using the messaging feature. I look forward to work with you in future.
Thank you!??
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
In countries with high birthrates, such as many of the Central and South American nations, unemployment rates are typically far higher than in countries with lower birthrates. Explain why.
-
Based upon the table provided. determine as many of the concrete mix design characteristics as possible for the following: ---- A structural concrete column within a school building that is 450x450...
-
a. Use the spreadsheet to calculate as many of the company's Profitability, Turnover-Control, and Leverage and Liquidity ratios as you can for these years (see Table 2.5 in text for a list of...
-
Describe, draw, the curve with the following parametric equations in the cylindrical co-ordinate space: r = 5, z = 2,0 = t
-
Refer to Example 13.9 and the data in CRIME4.RAW. (i) Suppose that, after differencing to remove the unobserved effect, you think log(polpc) is simultaneously determined with log(crmrte); in...
-
Vin Pinball Machines, Inc. makes two types of pinball machines: the SM pinball machine and the Zelda pinball machine. Vintage Pinball has the following unit-level data for the two products: How many...
-
The following financial data is taken from the annual report of The Emma Company: Explain (a) what other comprehensive income is and how it differs from net income, (b) what happened to the value of...
-
An analysis of transactions for Arthur Cooper & Co. was presented in Exercise. In Exercise, An analysis of the transactions made by Arthur Cooper & Co., a certified public accounting firm, for the...
-
System Operational Requirement: Provide a List of COI's and KPP's As a member of the TSA Homeland Security team is to generate a list of "Critical Operational Issues" (COI) and then provide a list of...
-
Table 2.9 gives data on mean Scholastic Aptitude Test (SAT) scores for collegebound seniors for 19722007. These data represent the critical reading and mathematics test scores for both male and...
-
Name or draw the following compounds. (a) cis-2,3-Diphenyloxacyclopropane; (b) 3-azacyclobutanone; (c) 1,3-oxathiacyclopentane; (d) 2-butanoyl-1,3-dithiacyclohexane; (a) (b) (c) (d)
-
Identify by name (either IUPAC or common) as many of the heterocyclic rings contained in the structures shown in Table 25-1 as you can. Top Ten U.S. Prescription Drugs (Ranked by 2007 Sales) by...
-
Critically discuss the effects of Marijuana. Outline a comparision of Marijuana Societal Costs vs. Business Benefits
-
Which of the following best describes the reason why an independent auditor reports on financial statements? a. A management fraud will be detected by independent auditors. b. Different interests may...
-
A hypothesis test is performed at a significance level = 0.05. What is the probability of a Type I error?
-
The independent auditor lends credibility to client financial statements by a. Stating that the financial statements are stated in accordance with generally accepted auditing standards. b....
-
Which of the following types of audits is performed to determine whether a person or entity has adhered to policies, laws, and regulations? a. Operational audit. b. Compliance audit. c. Financial...
-
Discuss the effect of mismatched transistors on the characteristics of the MOSFET two-transistor current source.
-
How can cohesiveness and diversity support group effectiveness?
-
a. Why does the Wi-Fi Alliance release compatibility testing profiles in waves instead of combining the entire standards features initially? 27a1.) An 802.11ac Wi-Fi compatibility testing profile...
-
Meerwein?s reagent, triethyloxonium tetra-fluoroborate, is a powerful ethylating agent that converts alcohols into ethyl ethers at neutral pH. Show the reaction of Meerwein?s reagent with...
-
Safrole, a substance isolated from oil of sassafras, is used as a perfumery agent. Propose a synthesis of safrole from catechol (1,2-benzenediol). CH2CH=CH2 Safrole
-
Epoxides are reduced by treatment with lithium aluminum hydride to yield alcohols. Propose a mechanism for thisreaction. 1. LIAIH4, ether 2. H*
-
A beverage manufacturer pays a flat fee of $100 per month to have access to city water. In addition, the manufacturer pays $5 per every 10,000 gallons used. In what way should this cost be...
-
The following media statement was released by a director of a company listed on the JSE Limited: "Our assets have almost doubled in value in the last two years. Anyway, who cares about anything else...
-
Suppose you buy both a $ 1 , 0 0 0 , 5 % annual coupon bond with a maturity of 8 years and a zero coupon bond with a maturity of 8 years. The yield is 6 . 2 5 % when you buy them, but changes to 6 %...

Study smarter with the SolutionInn App


