Predict the spin spin splitting that you would expect to observe in the NMR spectra of
Question:
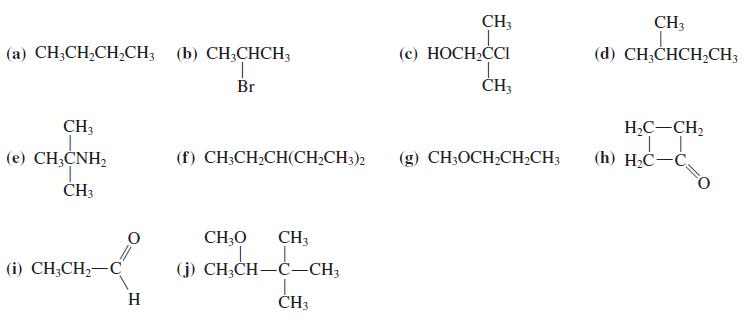
Predict the spin – spin splitting that you would expect to observe in the NMR spectra of each compound in Problem 36. (Reminder: Hydrogens attached to oxygen and nitrogen do not normally exhibit spin – spin splitting.)
Transcribed Image Text:
CH3 CH3 (а) СН,СH,CH,CH; (с) НОСН,СCI (d) CH;CHCH,CH3 (b) CH;CHCH; 1. Br ČH; CH; H2C-CH2 (е) CН,CNH, (f) CH;CH2CH(CH;CH3)2 (g) CH;OCH,CH2CH; (h) H-С—С. ČH3 CH;O CH3 (i) CH;CH,-C (j) CH;CH-C-CH3 H ČH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
SNo spinspin splitting a C1triplet3H C2quartet2H b C1doublet6H C2septet1H c O...View the full answer

Answered By

Bharat Singh Patel
I like teaching since my matriculation. That time i was teaching 6-8 class students with all subjects. I was very interested in teaching. Then after i have done my 10+2. I joined a coaching class. Where i was teaching mathematics and chemistry of 10th class student. After that i moved to Ewing Christian College, Allahabad for Graduation. then i taught chemistry only. then I clear IIT JAM to got admission in IIT Jodhpur for M.Sc. During M.Sc I have cleared CSIR-JRF With AIR 54 and GATE with AIR 114. In college time I always cleared doubts of classmates and juniors.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Name one element that you would expect to exhibit bonding properties similar to boron. Explain?
-
Give the products that you would expect to obtain when the following compounds are treated with ozone, followed by work-up with a. Zn, H2O: b. H2O2 1. 2. CH2¡CHCH2CH2CH2CH3 3. 4. 5....
-
Identify two types of companies that you would expect to have a high gross profit margin and two types of companies that you would expect to have a low gross profit margin.
-
On December 1, 2018, BEEN RICHARDS Company entered into two independent forward contracts to sell US$1,200 in 90 days or on March 1, 2019. The exchange rates available on various dates are as...
-
What does procurement mean?
-
10) A firm's market value balance sheet shows cash of $400,000, other assets of $450,000, and equity of $850,000. The firm has 6,000 shares of stock outstanding and will repurchase 1,000 shares of...
-
While designing a mixer-settler extraction system, you obtain a mass transfer correlation from a book. Unfortunately, the book does not explain which model was used. Which model would you use to...
-
Artis owns 40%of the Rhode Island Chile Parlor (RICP). During the current year, Rhode Island gives Artis fringe benefits worth $4,000 in addition to his $30,000 salary. RICP's net taxable income...
-
Francis and Hartley Law Office employs 12 full-time attorneys and 10 paraprofessionals. Direct and indirect costs are applied on a professional labor-hour basis that includes both attorney and...
-
Thomas Ncube is a state-registered nurse at a nearby local Polyclinic. He has maintained a satisfactory account with your branch for the past five years, with occasional overdrafts prior to receipt...
-
For each compound in each of the following groups of isomers, indicate the number of signals in the 1 H NMR spectrum, the approximate chemical shift of each signal, and the integration ratios for the...
-
Predict the spin spin splitting that you would expect to observe in the NMR spectra of each compound in Problem 37. CH3 CH3 CH3 (a) CH;CCH,CH3, BICH,CHCH,CH3, CH;CHCH,CH;Br Br CH,CI CH; (b)...
-
For which of these events are the outcomes mutually exclusive? Circle "Yes" if the outcomes are mutually exclusive or "No" if the outcomes are not mutually exclusive. Event Mutually Exclusive? a....
-
What is an SSR and what is it intended to accomplish?
-
Create a simple WBS for planning a wedding.
-
What would it mean if a difference were statistically significant but the effect size were not meaningful?
-
What is an HSR and SSR and what is it intended to accomplish?
-
What is verification, what is its primary objective, and what constitutes Verification success?
-
(a) From the plot of yield strength versus (grain diameter)-1/2 for a 70 Cu-30 Zn cartridge brass in Figure 7.15, determine values for the constants 0 and ky in Equation 7.7. (b) Now predict the...
-
Stephen Schor, an accountant in New York City, advised his client, Andre Romanelli, Inc., to open an account at J. P. Morgan Chase Bank, N.A., to obtain a favorable interest rate on a line of credit....
-
In an experiment to measure the molar mass of a gas, 250 cm3 of the gas was confined in a glass vessel. The pressure was 152 Torr at 298 K and, after correcting for buoyancy effects, the mass of the...
-
A certain sample of a gas has a volume of20.00 dm ' at OCand 1.000 atm. A plot of the experimental data of its volume against the Celsius temperature, , at constant p, gives a straight line of slope...
-
A certain gas obeys the van der Waals equation with a =0.76 m6 Pa mol-2, its volume is found to be 4.00 X 10-4 m3 mol-1 at 288 K and 4.0 MPa. From this information calculate the van der Waals...
-
39.A firm's free cash flow in 2020 (FCF 0 ) was $180 million, and expect its free cash flow has a constant growth rate at 5%. If the weight average cost of capital (WACC) for the firm is 10%, what's...
-
Efram and Sofia, both aged 2 4 , are newlyweds. Their after - tax salaries are $ 5 5 , 0 0 0 and $ 4 9 , 0 0 0 , respectively. They have been renting a flat since they started work but have decided...
-
rewrite the following for clarity and justification: Inventory analysis and reporting on each stock-keeping unit (SKU) are crucial to effectively managing lead times

Study smarter with the SolutionInn App


