Reductive amination of excess formaldehyde with a primary amine leads to the formation of a dimethylated tertiary
Question:
Reductive amination of excess formaldehyde with a primary amine leads to the formation of a dimethylated tertiary amine as the product (see the following example). Propose an explanation.
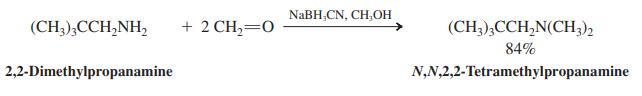
Transcribed Image Text:
NABH,CN, CH,OH (CH3),CCH,NH, + 2 CH,=0 (CH3),CCH,N(CH;), 84% 2,2-Dimethylpropanamine N,N,2,2-Tetramethylpropanamine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 28% (7 reviews)
The given reaction is reductive amination or reductive alkylation When 22 dimethylpro...View the full answer

Answered By

User l_359487
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Heating acetone with sulfuric acid leads to the formation of mesitylene (1, 3, 5-trimethylbenzene). Propose a mechanism for this reaction.
-
The two most general amine syntheses are the reductive amination of carbonyl compounds and the reduction of amides. Show how these techniques can be used to accomplish the following syntheses. (a)...
-
Excess ammonia must be used when a primary amine is synthesized by reductive amination. What product will be obtained if the reaction is carried out with an excess of the carbonyl compound instead?
-
A student has six textbooks, each with a thickness of 4.0 cm and a weight of 30 N. What is the minimum work the student would have to do to place all the books in a single vertical stack, starting...
-
A country has had a steady value for its floating exchange rate (stated inversely as the domestic currency price of foreign currency) for a number of years. The country now tightens up on (reduces)...
-
Firm C is considering the acquisition of Firm T. Firm C has estimated the cash flows, cost of capital, and growth rate for firm T shown below. Answer the following questions to estimate the current...
-
The Mann Corporation began operations in 2015. Information relating to the companys purchases of inventory and sales of products for 2015 and 2016 is presented below. Calculate the LIFO cost of goods...
-
Preparing the income statement and balance sheet using the accrual basis Bob Hansen opens a retail store on January 1, 2008. Hansen invests $50,000 for all of the common stock of the firm. The store...
-
An employee terminated for violating a company policy threatened to sue the company for wrongful termination. The policy that the employee violated was the company's password disclosure policy, which...
-
Figure 4-32 shows a class list for Millennium College. Convert this user view to a set of 3NF relations using an enterprise key. Assume the following: ¢ An instructor has a unique location....
-
Reductive amination of excess formaldehyde with a primary amine leads to the formation of a dimethylated tertiary amine as the product (see the following example). Propose an explanation. NABH,CN,...
-
Several of the natural amino acids are synthesized from 2-oxocarboxylic acids by an enzymecatalyzed reaction with a special coenzyme called pyridoxamine. Use electron-pushing arrows to describe each...
-
On March 1, 2010, Dora Corporation began operations with a charter it received from the state that authorized 50,000 shares of $4 par value common stock. Over the next quarter, the company engaged in...
-
When the number of degrees of freedom is large, the Students t distribution is close to the ______________ distribution. In Exercises 7 and 8, fill in each blank with the appropriate word or phrase.
-
Level 95% In Exercises 2528, find the critical value z2 needed to construct a confidence interval with the given level.
-
Under limited circumstances, CPAs may express unqualified opinions on financial statements that utilize accounting principles other than those for- mulated by designated bodies. Give two examples of...
-
Are analytical procedures very effective for discovering errors and frauds?
-
How do these standards differ for (1) errors, (2) frauds, (3) direct-effect illegal acts, and (4) far-removed-effect illegal acts?
-
Describe the strategic management process and SWOT analysis.
-
Suppose Green Network Energy needs to raise money to finance its new manufacturing facility, but their CFO does not think the company is financially capable of making the periodic interest payments...
-
Show syntheses of these compounds from(Z)-2-butene: ) - a) H H C-C - CH3 . .
-
Show the products of thesereactions: . C-C CH3 1) O, CH,OH 1) O3. CH,OH a) CH;CH,CH-CH2 b) 2) (CH,),S 2) (CH,)2S CH3 . 1) 0,, CH;OH 1) 0,, CH,OH d) 2) (CH3),S 2) (CH3)2S 1) O3, CH,OH 2) (CH3),S
-
Show the alkenes that produce these compounds onozonolysis: a) CH,CCH,CH, + CH,CH b) c) CH;CCH,CH;CH + HCH + CH,CH
-
Intro You have collected the following information about a company: Source of capital Market value Book value After-tax cost Long-term debt 100,000 100,000 0.08 Preferred stock 60,000 72,000 0.11...
-
IV (Mortgage-Backed Securities) 15 points You purchase an MBS 9, 30 year with a face value of $100,000 a) What is your monthly payment? b) What is the interest and principal earned over 5 years c)...
-
Based on the following information, what is the expected return? (Input decimal, keep 4 digits. For example, if you get 7.56%, then input 0.0756.) Probability of State of Rate of Return if State of...

Study smarter with the SolutionInn App


