Suggest a good synthetic method for preparing each of the following haloalkanes from the corresponding alcohols. CH3
Question:
Suggest a good synthetic method for preparing each of the following haloalkanes from the corresponding alcohols.
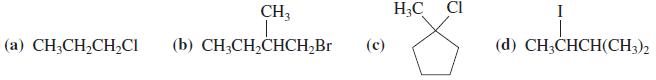
Transcribed Image Text:
CH3 H3C CI I (а) CH-CH-CH,CI (b) CH;CH,CHCH,Br (с) (d) CH;CHCH(CH3)2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
3 ...View the full answer

Answered By

Yadram Dhanka
I was engaged in conducting private tuitions for students of class 11th and 12th. I would like to work with a leading educational organization and to use my in-depth subject knowledge and passion towards teaching to the best of my ability, so as to enrich the student’s ability to learn, as well as to advance my career in the education sector.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Outline the best method for preparing each of the following compounds from an appropriate alcohol. (a) (b) CH3CH2CH2CH2COOH (c) (d) (e) , 0 CH CH
-
Suggest a sequence of reactions suitable for preparing each of the following compounds from the indicated starting material. You may use any necessary organic or inorganic reagents. (a) 1-Propanol...
-
Suggest appropriate methods for preparing each of the following compounds from the starting material of your choice. (a) CH3CH2CH2CH2CH2MgI (c) CH3CH2CH2CH2CH2Li (b) CH3CH2CCMgI (d)...
-
Refer to the GrandScapes data set. Requirements 1. Compute the direct labor rate variance and the direct labor efficiency variance. 2. What is the total variance for direct labor? 3. Who is generally...
-
Explain why the buyer has more risk in a time and materials contract than in a fixed-price contract.
-
A2 - capacitor initial battery I I I- capacitor (with dielectric) I- - Step 1 remove dielectric Step 2 disconnect battery Step 3 - bring plates closer Initially, a parallel-plate capacitor is...
-
How should the leaving chief executive be involved in the succession planning and transition process?
-
Determine the number of cycles per day and the production quantity per cycle for this set of vehicles: Product Daily Quantity A ......... 21 B ......... 12 C ......... 3 D ......... 15 Use the...
-
Adra is learning English vocabulary in school. If Adra studies her vocabulary list for 15 minutes everyday for a week she performs much better than if she's crams for 2 hours the night before. This...
-
Selected comparative financial statement information of Bluegrass Corporation follows. Required 1. Compute each years current ratio. Round ratios to one decimal. 2. Express the income statement data...
-
Give the expected product(s) of the reaction of trans-3-methylcyclopentanol with each of the reagents in Problem 38. (a) K +- OC(CH 3 ) 3 (b) Sodium metal (c) CH 3 Li (d) Concentrated HI (e)...
-
Suggest the best syntheses for each of the following ethers. Use alcohols or haloalkanes or both as your starting materials. () (b) () (d) () (f)
-
Find the fractions equal to the given decimals. 0.1181818 . . .
-
What is an N2 Diagram? What is its purpose and how is it used? What problem(s) does it solve for an SE?
-
What is the Developmental Configuration?
-
What is a Context Diagram? What is its purpose and how is it used?
-
What are the workflow sequence steps in System Development Phase?
-
What is the 100X Software Rule and what are its implications to System Development?
-
Determine whether it is possible to cold work steel so as to give a minimum Brinell hardness of 240 and at the same time have a ductility of at least 15%EL. Justify your answer.
-
If the amplifier indicated by the box input impedance of oo, which of the following statements are true ? has an open loop gain as well as Feedback factor (\beta = 1/ R_1\) The feedback is voltage...
-
The molar mass of an enzyme was determined by dissolving it in water, measuring the osmotic pressure at 20C, and extrapolating the data to zero concentration. The following data were obtained: c/(mg...
-
Given that p*(HzO) = 0.02308 atm and p (HzO) = 0.02239 atm in a solution in which 0.122 kg of a non-volatile solute (M = 241 g mol-1) is dissolved in 0.920 kg water at 293 K, calculate the activity...
-
Suppose the incident sunlight at ground level has a power density of 0.87 kW m-2 at noon. What is the maximum rate of loss of water from a lake of area 1.0 ha? (1 ha = 104 m2.) Assume that all the...
-
20. The employees of Book Nook paid a total of $2178.40 in Social Security tax last month, $509.46 in Medicare tax, and $5583.19 in federal withholding tax. Find the total amount that the employer...
-
For what type of client are fee-based accounts most appropriate? For those who need a full suite of financial planning services. For those who trade actively and want to save on commissions. For...
-
How does theses four concepts relate to each other ( MPS, MRP, Time fences, ERP)

Study smarter with the SolutionInn App


