Write the expected major product of reaction of each of the carbonyl compounds (i) (iii) with
Question:
Write the expected major product of reaction of each of the carbonyl compounds (i) – (iii) with each of the reagents (a) – (h).
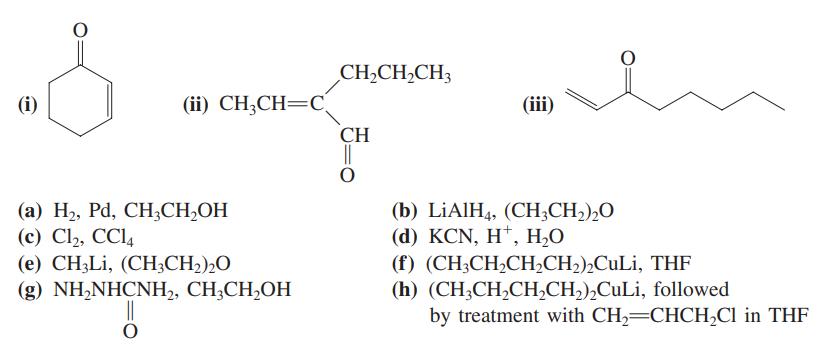
Transcribed Image Text:
CH2CH,CH3 (i) (ii) CH;CH=C. (iii) CH || (а) Н, Pа, СН,CН-ОН (c) Cl2, CCl, (e) CH;Li, (CH3CH2),O (b) LİAIH4, (CH;CH,),O (d) KCN, H*, Н.О (f) (CH;CH,CH,CH2),CuLi, THF (h) (CH3CH,CH,CH2),CuLi, followed by treatment with CH2=CHCH,Cl in THF (g) NH,NHCNH,, CH,CH,ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Write the expected major product of reaction of 1-propynyllithium, CH 3 C C - Li + , with each of the following molecules in THF. CI () C,CH,Br (b) () clohexanone CH3 (d) -H () C,CHH (f)
-
Write the expected major product(s) of each of the following attempted ether syntheses, (a) (b) (c) (d) (e) (f) DMSO CH,CH CHCI CH CH CHCH CH Cl HMPA CH CH CHO +CH,CH CHCH CH .. DMSO + CH3I (CH)...
-
Give the expected major product of reaction of 2,2-dimethyloxacyclopropane with each of the following reagents? (a) Dilute H2SO4 in CH3OH (b) Na+ -OCH3 in CH3OH (c) Dilute, aqueous HBr (d)...
-
If a particular glucose fermentation process is 87.0% efficient, how many grams of glucose would be required for the production of 51.0 g of ethyl alcohol (C 2 H 5 OH)? C 6 H 12 O 6 2C 2 H 5 OH +...
-
Explain how section 1139 found in Figure 4-2 might be used in the Welsh case.
-
On the planet of Mercury, 4-year-olds average 3.1 hours a day unsupervised. Most of the unsupervised children live in rural areas, considered safe. Suppose that the standard deviation is 1.3 hours...
-
Describe blueprinting.
-
Use of materials and manufacturing labor variances for benchmarking, you are a new junior accountant at Clear view Corporation, maker of lenses for eyeglasses. Your company sells generic-quality...
-
Case: Pharmacy Service Improvement at CVS(A) 1. Developing on the problematization presented in the case, design an information system that can potentially solve the problems facing CVS. 2. Looking...
-
1. Compare the leadership traits and behaviors of Ben Samuels and Phil Jones. 2. Which leader do you think is more effective? Why? Which leader would you prefer to work for? 3. If you were Phil Jones...
-
The distillate from sandalwood is one of the oldest and most highly valued fragrances in perfumery. The natural oil is in short supply and, until recently, synthetic substitutes have been difficult...
-
Give the expected product(s) of each of the following reactions. 1. LDA, THF 2. BICH,COCH, H 1. LDA, THF 2. CH.CH Br, HMPA (a) CH,CCH,CH,CH3 (b) 0:
-
Myssie Cardenas was recently hired as the chief financial officer for Barajas Corporation. At the time Myssie was hired, the company had just completed the accounting cycle for the year ending...
-
What is the applicability of the Rules of Conduct to the practice of public accounting in the United States?
-
Find the z-scores that bound the middle 80% of the area under the standard normal curve.
-
a. What is the essence of Rule 301-Confidential Client Information? b. Indicate three exceptions to this rule.
-
Enumerate the specific parts of Rule 201-General Standards.
-
a. What conditions must be met to issue a report on internal accounting control as a by-product of an audit engagement? b. Indicate the essential points to be covered in reporting to a governmental...
-
An effective evaluation should accomplish at least three things. Name them.
-
Charles owns an office building and land that are used in his trade or business. The office building and land were acquired in 1978 for $800,000 and $100,000, respectively. During the current year,...
-
Azurin is a protein containing a copper ion that shuttles between the +2 and +1 oxidation states, and cytochrome c is a protein in which a haem-bound iron ion shuttles between the +3 and +2 oxidation...
-
What pressure of nitrogen gas is required to produce a collision rate of 5.00 x 1019 S-1 at 525 K on a circular surface of diameter 2.0 mm?
-
Calculate the average rate at which He atoms strike an iron atom in a surface formed by exposing a (l00) plane in metallic iron to helium gas at 100 K and a pressure of24 Pa. Crystals of iron are...
-
This is what I need but with these variables: Data Collected ( in dollars ) Like Games Our Play Industry Average Accounts receivable 5 , 4 0 0 7 , 8 0 0 7 , 7 0 0 Net fixed assets 1 1 0 , 0 0 0 1 6 0...
-
Organisational procedures state that all corporate bookings made through SJ Travel incur a 10% mark up to increase profit margins. Show the calculation to mark up a gross fare of $1000.00 by 10% to...
-
all the below calculations should be shown on excel spreadsheet

Study smarter with the SolutionInn App


