Write the expected product(s) of each of the following reactions. CH3 3 NANH, liquid NH, 2 NANH,,
Question:
Write the expected product(s) of each of the following reactions.
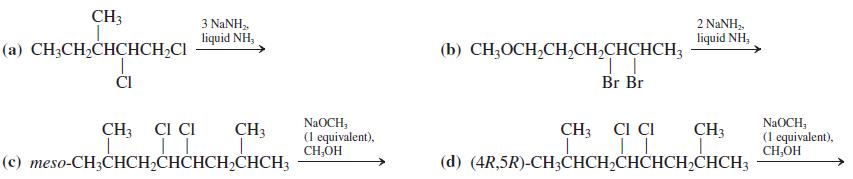
Transcribed Image Text:
CH3 3 NANH, liquid NH, 2 NANH,, liquid NH, (а) CH-CH-CHCНCH-CI (b) CH,OCH,CH,CH,CHCHCH3 ČI Br Br NaOCH, (1 equivalent), CH,OH CH3 CI CI NaOCH, (1 equivalent), CH,OH CH3 CI CI CH3 CH3 (с) тeso-CH;CHCH-CНCHCH,CHСH; (d) (4R,5R)-CH;CHCH,CHČHCH2CHCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the major product(s) of each of the following reactions.
-
Give the major product(s) of each of the following reactions.
-
Give the major product(s) of each of the following reactions. (a) (b) (c) (d) (e) (f) H;C I HO H
-
The net present value and internal rate of return desirability measures for two mutually exclusive investments being considered by Stockton Corporation to follow. Year NPV IRR R 161 14.60% S 138...
-
Explain timing and accuracy of the three levels of cost estimates: ROM, budget, and definitive.
-
A company uses a job cost system. As of January 1, its records showed the following balances: Materials Work in Process Finished Goods (Job No. 211) 13,500 25,800 30,000 The work in process inventroy...
-
The Apollo Company is a catalogue-based retailer. The following describes Apollos operations in its first two years of business: Required 1. Prepare an income statement for each year assuming that...
-
Ritz Products materials manager, Bruce Elwell, must determine whether to make or buy a new semiconductor for the wrist TV that the firm is about to produce. One million units are expected to be...
-
What kind of factors influences a person's ability to solve a problem? What are some examples of problems that people have a hard time solving, and why? What are some ways that you could improve your...
-
Use information on the endpapers of this book to calculate the average density of the Earth. Where does the value fit among those listed in Tables 1.5 and 14.1? Look up the density of a typical...
-
The IR spectrum of 1,8-nonadiyne displays a strong, sharp band at 3300 cm -1 . What is the origin of this absorption? Treatment of 1,8-nonadiyne with NaNH 2 , then with D 2 O, leads to the...
-
(a) Write the expected product of the reaction of 3-octyne with Na in liquid NH 3 . (b) When the same reaction is carried out with cyclooctyne (Problem 33b), the product is cis-cyclooctene, not...
-
Describe and demonstrate approaches to planning, running, and following up on meetings.
-
What factors should the auditor consider when developing specific audit objectives? Can there be more than one specific audit objective for a particular account for each category of assertion?...
-
Why is it important to have an adequate balance of each of these factors (cited in your response to question 2) contributing to personal security?
-
What are the purposes of an engagement letter?
-
Describe three types of information that an auditor might obtain to gain an understanding of the entitys business and industry. How might these three types of information affect the audit plan?
-
What are the important sources of personal security? Explain each one briefly.
-
Indium atoms are to be diffused into a silicon wafer using both pre-deposition and drive-in heat treatments; the background concentration of In in this silicon material is known to be 2 ( 1020...
-
Why do bars offer free peanuts?
-
The Humphreys series is a group of lines in the spectrum of atomic hydrogen. It begins at 12 368 nm and has been traced to 3281.4 nm. What are the transitions involved? What are the wavelengths of...
-
The Li2+ion is hydrogenic and has a Lyman series at 740 747 cm3, 877 924 cm-1, 925 933 cm-1, and beyond. Show that the energy levels are of the form -hcRln2 and find the value of R for this ion. Go...
-
W.P. Wijesundera, S.H. Vosko, and F.A. Parpia (Phys. Rev. A 51, 278 (1995)) attempted to determine the electron configuration of the ground state of lawrencium, element 103. The two contending...
-
A firm has 10,000,000 shares outstanding with a price per share of $22.70 (previous to a share repurchase). The firm repurchases 2,000,000 shares with a price per share of $26.90. A share repurchase...
-
Ailing Company's liabilities exceed its assets. Ailing hires Brad, an accountant, to certify a balance sheet showing a positive net worth. Credit Bank relies on the balance sheet to make a loan to...
-
Sharon owns stock that she purchased at various times. Based on the purchase and sales dates below, determine her long-term capital gain. Date Purchased Date Sold Purchase Price Sales Price 1/10/2023...

Study smarter with the SolutionInn App



