Write the final products of the following reaction sequences. (a) (b) (c) (d) Write a detailed mechanism
Question:
Write the final products of the following reaction sequences.
(a)
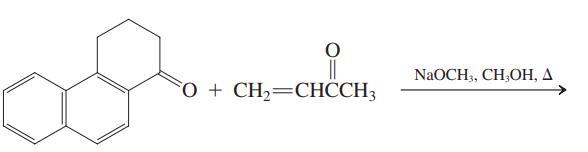
(b)
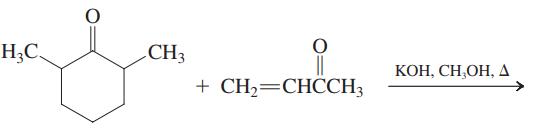
(c)
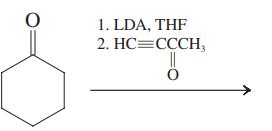
(d) Write a detailed mechanism for reaction sequence (c).
Transcribed Image Text:
NaOCH, CH,OН, Д O + CH2=CHCCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
a b c d HC S CH3 1 LDA THF CH LDA T...View the full answer

Answered By

Jatin Verma
Life is a chemical reaction, it only requires balancing! To balance your minds with my more than six years of experience I Jatin Verma here for all you better things for better living through chemistry and also to solve mysteries in chemistry I as your chemistry educator without even touching your arm it will set fire to your minds.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the product(s) of the following reaction sequences. 1. , * 2. 1. , * 2. (CH),CH.CI 3. ', . CH,Br 3. H., . () CH,CO (b) CH2CHO
-
Write a detailed mechanism for the following reaction. HBr (excess) 2 Br
-
Write a detailed mechanism for the following reaction. Br OH HBr
-
The B.B. Lean Co. has 1.4 million shares of stock outstanding. The stock currently sells for $20 per share. The firm's debt is publicly traded and was recently quoted at 93 percent of face value. It...
-
In re Marriage of Modnick, 33 Cal. 3d 897, 191 Cal. Rptr. 629, 663 P.2d 187 (1983)
-
3.15. Solution concentration. A chemist studied the concentration of a solution (Y) over time (X). Fifteen identical solutions were prepared. The 15 solutions were randomly divided into five sets of...
-
Describe the purpose of the internal audit function.
-
A well-respected polling agency has conducted a poll for an upcoming Presidential election. The polling agency has taken measures so that its random sample consists of 50,000 people and is...
-
Explain the differences between "IT Management" and "Enterprise Governance of IT"; and provide examples of organizational activities specific to Enterprise Governance of IT.
-
Brothers Mike and Tim Hargenrater began operations of their tool and die shop (H & H Tool, Inc.) on January 1, 2019. The annual reporting period ends December 31. The trial balance on January 1,...
-
Write the products of each of the following reactions after aqueous work-up. (e) Write the results that you expect from base treatment of the products of reactions (c) and (d). LDA, THE (a) CH;CCH; +...
-
Propose syntheses of the following compounds by using Michael additions followed by aldol condensations (i.e., Robinson annulation). Each of the compounds shown has been instrumental in one or more...
-
Evaluate the following expression: 7 + 3 * 2 > 6 * 3 AndAlso True. a. True b. False
-
Find the area under the standard normal curve to the left of a. z = 0.74 b. z = 2.16 c. z = 1.02 d. z = 0.15
-
What conclusions do you draw about (a) yourself as a consumer, and (b) the performance of each of the businesses in Exercise 1? Assess whether any of these businesses managed to develop a sustainable...
-
Describe the responsibilities and primary functions of the Division of Corporation Finance.
-
Identify the five divisions of the SEC.
-
Briefly describe the SEC's rules concerning (a) qualifications of independent accountants and (b) accourtant's reports.
-
Describe the two behaviors used in the Kelley Model and the resulting five follower types.
-
Wholesalers Ltd. deals in the sale of foodstuffs to retailers. Owing to economic depression, the firm intends to relax its credit policy to boost productivity and sales. The firms current credit...
-
For how long on average would an atom remain on a surface at 400 K if its desertion activation energy were? (a) 20 kO mol-1, (b) 200 k] mol-I? Take TO = 0.12 ps. For how long on average would the...
-
A solid in contact with a gas at 8.86 kPa and 25C adsorbs 4.67 mg of the gas and obeys the Langmuir isotherm. The enthalpy change when 1.00 mmol of the adsorbed gas is desorbed is +12.2 J. What is...
-
Suppose it is known that ozone adsorbs on a particular surface in accord with a Langmuir isotherm. How could you use the pressure dependence of the fractional coverage to distinguish between...
-
Jamie Lee and Ross, now 57 and still very active, have plenty of time on their hands now that the triplets are away at college. They both realized that time has just flown by, over twenty-four years...
-
Line Item Amount ($) 1. Estimated Revenues To be determined 2. Estimated Expenses 119,108.03 3. Itemized Direct Costs 77,108.03 (Training Material: 10,000, Salary/Wages: 62,108.03, Venue Rental:...
-
2) The risk free rate of interest is 3.0%. Inflation is expected to be 3.0% this year, 2.8% next year 2.5% the following year and 2.3% in each of the following years. Assume the liquidity premium is...

Study smarter with the SolutionInn App


