Find the frequency of the lowest vibrational mode of a diatomic molecule in terms of the parameters
Question:
Find the frequency of the lowest vibrational mode of a diatomic molecule in terms of the parameters of the Morse potential, eq.
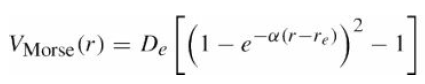
Transcribed Image Text:
Vstone () = De (1-e -r)* -1 -a(r-re
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
Expand V Morse r to second order about its minimum First fi...View the full answer

Answered By

Sumit kumar
I am an experienced online essay writer with a thorough understanding of any curriculum.and subject expert at Chegg for mathematics, CS subjects..
4.90+
5+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
The overall length of a piccolo is 32.0 cm. The resonating air column vibrates as in a pipe open at both ends. (a) Find the frequency of the lowest note that a piccolo can play, assuming that the...
-
For the arrangement shown in Figure P18.64, /θ = 30.0°, the inclined plane and the small pulley are frictionless, the string supports the object of mass M at the bottom of the plane, and...
-
Magneto plasma frequency use the method of Problem 5 to find the frequency of the uniform plasma mode of a sphere placed in a constant uniform magnetic field B. Let B he along the z axis. The...
-
Write the appropriate SQL statement for the following queries. The result of the queries will be checked from your computer. What privilege should a user be given to log on to the Oracle server? Is...
-
Two blocks are sliding to the right across a horizontal surface, as the drawing shows. In Case A the mass of each block is 3.0 kg. In Case B the mass of block 1 (the block behind) is 6.0 kg, and the...
-
Write a function that has this prototype: int replace(char * str, char c1, char c2); Have the function replace every occurrence of c1 in the string str with c2, and have the function return the...
-
Continuing the previous two problems, how much total interest will you pay throughout the life of this loan? a. $4,565.98 b. $6,886.73 c. $8,684.03 d. $9,632.28
-
The flowing jobs are waiting to be processed at Rick Carlsons machine center. Carlsons machine center has a relatively long backlog and sets fresh schedules every 2 weeks, which do not disturb...
-
Question 4 ind P(2) for P(x)=2x^(4)-2x^(3)-4x^(2)+7 the remainder for the associated division and the val
-
Record the following transactions in the basic accounting equation. Treat each one separately. Assets = Liabilities + Owners Equity 1. Morgan invests $124,000 in company. 2. Bought equipment for...
-
The power output of air conditioners is measured intons, an ancient nomenclature dating back to the days when air was cooled by blowing it over blocks of ice.A ton of air conditioning is defined to...
-
The Morse potential parameters for oxygen are given by D e = 5.211 eV, = 2.78 1, and re =1.207 . Using the result from the previous problem, estimate the energy necessary to excite the oxygen...
-
Burkhart Company manufactures a product that has a variable cost of $25 per unit. Fixed costs total $1,000,000, allocated on the basis of the number of units produced. Selling price is computed by...
-
Evaluate three ways to deal constructively with complaints and anger from fellow employees, management, and customers within an organization. Are these methods effective to resolve the issues?...
-
Sam and Sally want to invest $500,000 in new restaurant. They need to achieve a 10% Return on that investment annually. The restaurant has fixed costs of $5,000 per month. Their estimated variable...
-
Should Mavi build a house of brands by developing new brands, separate from the Mavi brand, to pursue either the premium or the fast-fashion spaces. Explain why? https: //hbsp.harvard.edu/tu/009333e9
-
You buy an 8% coupon paid annually, 10-year maturity bond when its annual yield to maturity is 9.9%. A year later you sell the bond when the annual yield to maturity is 19%. What is return over the...
-
Find out if the CRM system links well with commonly used technology models and business systems (MS office, or others) or if customized, less familiar programs/interfaces are required.
-
Figure Q18.11 shows points on several equipotential surfaces, with V A > V D . (a) A positive test charge starts at point A and moves to point C. If the test charge starts and ends at rest, which of...
-
An Atomic Energy Commission nuclear facility was established in Hanford, Washington, in 1943. Over the years, a significant amount of strontium 90 and cesium 137 leaked into the Columbia River. In a...
-
A uniform marble rolls without slipping down the path shown in Fig. P10.80, starting from rest. (a) Find the minimum height h required for the marble not to fall into the pit. (b) The moment of...
-
Tarzan has foolishly gotten himself into another scrape with the animals and must be rescued once again by Jane. The 60.0-kg Jane starts from rest at a height of 5.00 m in the trees and swings down...
-
A block with mass m is revolving with linear speed v 1 in a circle of radius r 1 on a frictionless horizontal surface (see Fig. E10.42). The string is slowly pulled from below until the radius of the...
-
hello , can you please help me answer those a question correctly, thank you. Saved Help Save & Exit Subn Keisha owns 100% of DEF Company and has a basis ofof $12,000. Keisha has a: $10,000 in its...
-
Accountants that are focused on issuing financial statements to serve the dicision making needs of external users of the business are called
-
Under the default provisions, the financial rights between member-management owners and manager-management owners is: Question 2 options: a) immaterial. b) material, giving a higher percentage of...

Study smarter with the SolutionInn App


