An electrochemical cell consists of a silver metal electrode immersed in a solution with [Ag + ]
Question:
An electrochemical cell consists of a silver metal electrode immersed in a solution with [Ag+] = 1.0 M separated by a porous disk from a copper metal electrode. If the copper electrode is placed in a solution of 5.0 M NH3 that is also 0.010 M in Cu(NH3)42+, what is the cell potential at 25οC?
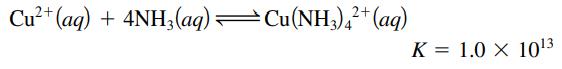
Transcribed Image Text:
Cu²+ (aq) + 4NH3(aq) —Cu(NH3)4²+ (aq) K = 1.0 X 10¹3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
The cell potential at 25C Ecell Ag10 M 00060 M Cu2aq50 M0010 ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
An electrochemical cell consists of a silver metal electrode immersed in a solution with [Ag + ] = 1.00 M separated by a porous disk from a compartment with a copper metal electrode immersed in a...
-
An electrochemical cell consists of a nickel metal electrode immersed in a solution with [Ni 2+ ] = 1.0 M separated by a porous disk from an aluminum metal electrode. a. What is the potential of this...
-
Gooran, Inc., has current assets of $240 million; property, plant, and equipment of $380 million; and other assets totaling $120 million. Current liabilities are $170 million and long-term...
-
How could a system be designed to allow a choice of operating systems from which to boot? What would the bootstrap program need to do?
-
The preparation of the sex pheromone of the bollworm moth, (E )-9,11-dodecadien-1-yl acetate, from compound A has been described. Suggest suitable reagents for each step in this sequence. Compound A...
-
Determine if 1,477 is prime.
-
For 2012, Eurie Company reported its most significant decline in net income in year. At the end of the year, H. Finn, the president, is presented with the following condensed comparative income...
-
At the end of 2022, the following information is available for Great Adventures. Additional interest for five months needs to be accrued on the $32,600, 6% loan obtained on August 1, 2021. Recall...
-
Use T-accounts to record the 4 months of transactions noted below for this new start-up company. Record all entries affecting the income statement into Equity since there are no separate T-accounts...
-
Cadmium sulfide is used in some semiconductor applications. Calculate the value of the solubility product constant (K sp ) for CdS given the following standard reduction potentials: CdS (s) + 2e ...
-
Consider a concentration cell that has both electrodes made of some metal M. Solution A in one compartment of the cell contains 1.0 M M 2+ . Solution B in the other cell compartment has a volume of...
-
Sunday, March 16, 2008, was not a peaceful day for the Board of Governors. Over the prior week, one of Wall Street s most famous investment houses, Bear Stearns, had gone into full collapse. Although...
-
During 2005 , Liberty's days' sales in receivables ratio was a. 39 day's b. 37 day's c. 35 day's d. 30 days
-
Liberty's inventory turnover during 2007 was a. 6 times b. 7 times c. 8 times d. Not determinable from the data given
-
Liberty's stock has traded recently around \(\$ 44\) per share. Use your answer to question 9 to measure the company's price/earnings ratio. a. 36 b. 44 c. 1.00 d. 69
-
Triumph Corporation is preparing its statement of cash flows by the indirect method. Triumph has the following items for you to consider in preparing the statement. Identify each item as an -...
-
Anita Maxwell Company expects the following for 2007: Net cash provided by operating activities of $150,000 Net cash provided by financing activities of $60,000 Net cash used for investing...
-
Describe the order in which different types of tax credits are applied to reduce a taxpayer's tax liability.
-
Differentiate. y = ln(3x + 1) ln(5x + 1)
-
A bowling ball has a weight of 12 lb and the length of the lane is approximately 60. feet. Treat the ball in the lane as a one-dimensional box. What quantum number corresponds to a velocity of 7.5...
-
For a particle in a two-dimensional box, the total energy eigenfunctions are a. Obtain an expression for E nx , n y in terms of n x , n y , a, and b by substituting this wave function into the two...
-
Consider the contour plots of Problem P15.17. a. What are the most likely area or areas Îx Îy to find the particle for each of the eigenfunctions of HË depicted in plots af? b. For...
-
Which of these are good ways to organize new emails in your inbox? Multiple select question. Place emails related to all of your classes in the education folder. Move emails that have been addressed...
-
Markets may have difficulty providing the proper quantity of a public good because a. individuals will tend to become free riders, and private firms will have difficulty generating enough revenue to...
-
Ensuring that a firm has sufficient cash available on a daily basis is part of: Multiple Choice Working capital management. Capital budgeting. Organizational structure. Business organization. Capital...

Study smarter with the SolutionInn App


