Determine the carburizing time necessary to achieve a carbon concentration of 0.45 wt% at a position 2-mm
Question:
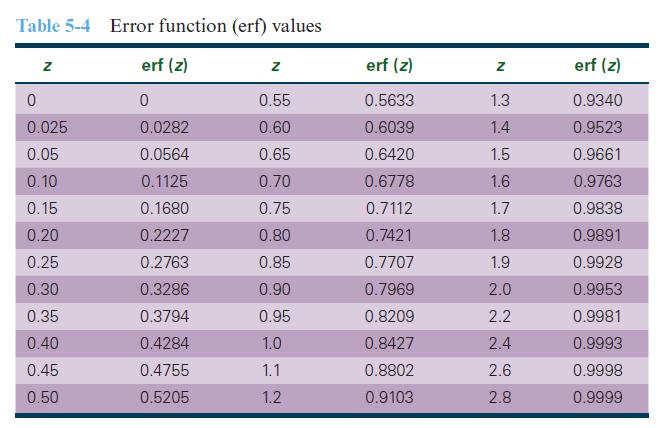
Determine the carburizing time necessary to achieve a carbon concentration of 0.45 wt% at a position 2-mm into a steel alloy that initially contains 0.20 wt% carbon. The surface concentration is to be maintained at 1.30 wt% at 1000°C. Use Table 5-4 for error function values as needed.
Transcribed Image Text:
Table 5-4 Error function (erf) values erf (z) 0 0.0282 0.0564 0.1125 0.1680 0.2227 0.2763 0.3286 0.3794 0.4284 0.4755 0.5205 Z 0 0.025 0.05 0.10 0.15 0.20 0.25 0.30 0.35 0.40 0.45 0.50 Z 0.55 0.60 0.65 0.70 0.75 0.80 0.85 0.90 0.95 1.0 1.1 1.2 erf (z) 0.5633 0.6039 0.6420 0.6778 0.7112 0.7421 0.7707 0.7969 0.8209 0.8427 0.8802 0.9103 Z 1.3 1.4 1.5 1.6 1.7 1.8 1.9 2.0 2.2 2.4 2.6 2.8 erf (z) 0.9340 0.9523 0.9661 0.9763 0.9838 0.9891 0.9928 0.9953 0.9981 0.9993 0.9998 0.9999
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 93% (15 reviews)
The carburizing time necessary is called formula3 and is g...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the carburizing time necessary to achieve a carbon concentration of 0.45 wt% at a position 2 mm into an iron-carbon alloy that initially contains 0.20 wt% C. The surface concentration is to...
-
Determine the carburizing time necessary to achieve a carbon concentration of 0.30 wt% at a position 4-mm into a steel alloy that initially contains 0.10 wt% carbon. The surface concentration is to...
-
Determine the carburizing time necessary to achieve a carbon concentration of 0.30 wt% at a position 4 mm into an iron-carbon alloy that initially contains 0.10 wt% C. The surface concentration is to...
-
A. Discuss the following conceptual model/framework of Orem's Self-Care Theory CONDITIONING FACTORS (12 MARKS) Orem's Self-Care Theory Conceptual Framework SELF-CARE AGENCY SELF-CARE DEFICIT NURSING...
-
What does limited liability mean?
-
Sean McNama is a renowned medical researcher at a large Canadian university. For many years he has been working on a technology that could significantly improve the vision of visually impaired...
-
Single product decision analysis Par-Tech, Inc. produces industrial cleaning com- pounds. One product group, especially good as a grease emulsifier, is called the Mol-Tech group. Three Mol-Tech...
-
CVP, target operating income, service firm Teddy Bear Daycare provides daycare for children Mondays through Fridays. Its monthly variable costs per child are: Teddy Bear charges each parent$600 per...
-
John purchased a stock that has a beta of 1.2, a standard deviation of 13%, and returned 16% this year. The market s return was 12% with a standard deviation of 14%. If the risk free rate of return...
-
Lolas operates a chain of sandwich shops. The company is considering two possible expansion plans. Plan A would open eight smaller shops at a cost of $8,550,000. Expected annual net cash inflows are...
-
Nitrogen from a gaseous phase is to be diffused into pure iron (BCC) at 700C. If the surface concentration is maintained at 0.10 wt% N, what will the concentration 1 mm from the surface be after 10...
-
Certain ceramics with special dielectric properties are used in wireless communication systems. Barium magnesium tantalate (BMT) and barium zinc tantalate (BZT) are examples of such materials....
-
Weiland Co. shows the following information on its 2016 income statement: sales = $173,000; costs = $91,400; other expenses = $5,100; depreciation expense = $12,100; interest expense = $8,900: taxes...
-
3. The income of B40 families has increased very slowly in the past one and two decades despite a variety of government policies to help the B40 families. One may, therefore, conclude that government...
-
Sunny Corporation has collected the following data for one of its products Direct materials standard (2 pounds per unit @ $0.57/lb.) Direct materials flexible budget variance - unfavorable Actual...
-
Bartlett Car Wash Company is considering the purchase of a new facility. It would allow Bartlett to increase its net income by $90,775 per year. Other information about this proposed project follows:...
-
Analyze how organizational purpose can empower a business strategy. Explain what a purpose-driven organization is and identify how it can lead to a competitive business strategy. analyze how state...
-
describe maybelline foundation finder. describe brand, packaging, quality,colours, variants and services. Describe the STAGE OF THE PRODUCT LIFECYCLE the product used and include facts that support...
-
A uniform rope of length L and mass M rests on a table. (a) If you lift one end of the rope upward with a constant speed, v, show that the rope's center of mass moves upward with constant...
-
Suppose that you could invest in the following projects but have only $30,000 to invest. How would you make your decision and which projects would you invest in? Project Cost $ 8,000 11,000 9,000...
-
Consider the following space-filling models for dry ice, ethanol, and caffeine: What amount (moles) is represented by each of the following samples? a. 1.50 g of dry ice b. 2.72 10 21 molecules of...
-
Prevacid is used to treat gastroesophageal reflux disease (GERD). The chemical formula of Prevacid is C 16 H 14 F 3 N 3 O 2 S. a. What is the molar mass of Prevacid? b. What mass of fluorine is in...
-
Freon-12 (CCl 2 F 2 ) is used as a refrigerant in air conditioners and as a propellant in aerosol cans. Calculate the number of molecules of Freon-12 in 5.56 mg of Freon-12. What is the mass of...
-
Business Planning The directors of Silicon Engineering Limited are concerned about the liquidity position of their company over the next three months. The following information is relevant: a ....
-
Prepare income statements for the company for each of its first two years under variable costing. ( Loss amounts should be entered with a minus sign. )
-
sury Stock Transactions and Presentation ) Clemson Company had the following stockholds of January 1 , 2 0 1 4 . \ table [ [ Common stock, $ 5 par value, 2 0 , 0 0 0 shares issued,$ 1 0 0 , 0 0 0

Study smarter with the SolutionInn App


