Determine the phases that are present and the compositions for each phase in Cu-85 wt% Ag at
Question:
Determine the phases that are present and the compositions for each phase in Cu-85 wt% Ag at 800°C.
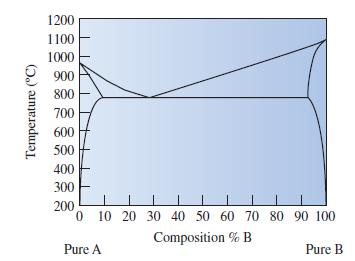
Transcribed Image Text:
Temperature (°C) 1200 1100 1000 900 800 700 600 500 400 300 200 0 10 20 30 40 50 60 70 80 90 100 Composition % B Pure A Pure B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
ANSWER At 800 C the phases present are liquid an...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the phases that are present and the compositions for each phase in Cu-55 wt% Ag at 600C. Data Form Problem 11-23 Consider a Pb-70% Sn alloy. Determine (a) If the alloy is hypoeutectic or...
-
Cite the phases that are present and the phase compositions for the following alloys: (a) 90 wt% Zn-10 wt% Cu at 400(C (750(F) (b) 75 wt% Sn-25 wt% Pb at 175(C (345(F) (c) 55 wt% Ag-45 wt% Cu at...
-
Cite the phases that are present and the phase compositions for the following alloys: (a) 90 wt% Zn-10 wt% Cu at 400C (750F) (b) 75 wt% Sn-25 wt% Pb at 175C (345F) (c) 55 wt% Ag-45 wt% Cu at 900C...
-
Consider a static (one-period), closed economy with one representative consumer, one rep- resentative firm, and a government. The level of capital K and government expenditures G in the economy are...
-
For the given of Problem 11, indicate: (a) How much would an interest arbitrageur earn if the foreign currency were at a forward premium of 1 percent per year? (b) What would happen if the foreign...
-
Cook Corporation reported the following related to property and equipment (all in millions): From the balance sheets: {Requirements} 1. Draw T-accounts for Property and Equipment and Accumulated...
-
What rules apply to service and filing of pleadings?
-
In what ways might a rms suppliers improve or undermine the rms Lean efforts? Can you think of any examples from the chapter that illustrate this idea?
-
Back in the day Circuit City was the big player in the electronic retail niche. It went belly up. Why did it fail when Best Buy Succeeded?
-
Harry Vincent, the Executive Vice President (EVP) of Monmouth, Inc. is considering acquiring a controlling stake (i.e., own more than 50% of shares) in Robertson Tool Company (RTC), a domestic...
-
A Pb-Sn alloy contains 45% and 55% at 100C. Determine the composition of the alloy. Is the alloy hypoeutectic or hypereutectic?
-
A Pb-Sn alloy contains 23% primary a and 77% eutectic microconstituent immediately after the eutectic reaction has been completed. Determine the composition of the alloy.
-
What is the structure of the beer industry?
-
Self-employed workers in the United States must pay Social Security taxes equal to 12.4% of any income up to \($118,500\) in 2015. This income level of \($118,500\) is known as the cap. Income in...
-
Suppose that the production function is Y = 9K 0.5 N 0.5. With this production function, the marginal product of labor is MPN = 4.5K 0.5 N -0.5 . The capital stock is K = 25 The labor supply curve is...
-
How strong must an electric field in a metal be in order for electrons in the field to have a drift speed of \(10 \mathrm{~mm} / \mathrm{s}\) if the time interval between electron-ion collisions is...
-
Air enters a pipe $(f=0.02)$ of diameter $0.05 \mathrm{~m}$ with stagnation pressure and temperature equal to $1 \mathrm{MPa}$ and $300 \mathrm{~K}$, respectively. The pipe exhausts into the ambient...
-
A copper wire that is \(600 \mathrm{~mm}\) long and has a radius of \(1.0 \mathrm{~mm}\) is connected to the terminals of a \(9.0-\mathrm{V}\) battery. What is the current through the wire...
-
On January 1 of this year, Barnett Corporation sold bonds with a face value of $500,000 and a coupon rate of 7 percent. The bonds mature in 10 years and pay interest annually on December 31. Barnett...
-
Interest Compounded Annually. When P dollars is invested at interest rate i, compounded annually, for t years, the investment grows to A dollars, where A = P(1 + i) t . Trevor's parents deposit $7800...
-
(a) Using only K sp from Table 6-3, calculate how many moles of Ca(OH) 2 will dissolve in 1.00 L of water. (b) How will the solubility calculated in part (a) be affected by the K 1 reaction in Table...
-
From the following equilibrium constants, calculate the equilibrium constant for the reaction HO,CCO,H 2H* + C,0.
-
Assuming complete dissociation of the salts, calculate the ionic strength of (a) 0.2 mM KNO 3 ; (b) 0.2 mM Cs 2 CrO 4 ; (c) 0.2 mM MgCl 2 plus 0.3 mM AlCl 3 .
-
The froghopper Philaenus spumarius is supposedly the best jumper in the animal kingdom. To start a jump, this insect can accelerate at 4.00 km/s over a distance of 2.0 mm as it straightens its...
-
find T(625). I Given the recurrence relation T(n)=7T (n/5)+ 10n for n > 1 T (1)=1 Answer: (please write your answer here, add required space if needed)
-
Given a string length of 1.00 0.01 m and a period of 2.00 0.10, what is the uncertainty in gravity? No need to include units, but give them as m/s^2.

Study smarter with the SolutionInn App


