Determine the temperature at which 10 percent of diatomic hydrogen (H 2 ) dissociates into monatomic hydrogen
Question:
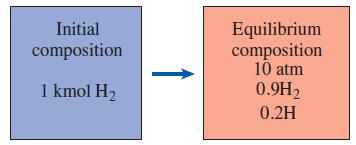
Determine the temperature at which 10 percent of diatomic hydrogen (H2) dissociates into monatomic hydrogen (H) at a pressure of 10 atm.
Transcribed Image Text:
Initial Equilibrium composition 10 atm 0.9H2 composition 1 kmol H2 0.2H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
The temperature at which 10 percent of H 2 dissociates into 2H is to be determined Assumptions 1 The ...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Determine the temperature at which 5 percent of diatomic oxygen (O2) dissociates into monatomic oxygen (O) at a pressure of 3 atm.
-
Determine the temperature at which 5 percent of diatomic oxygen (O2) dissociates into monatomic oxygen (O) at a pressure of 3 atm.
-
A mixture of 1 mol of H2 and 1 mol of Ar is heated at a constant pressure of 1 atm until 15 percent of H2 dissociates into monatomic hydrogen (H). Determine the final temperature of the mixture.
-
101, 115, 143, 106, 100, 142, 157, 163, 155, 141, 145, 153, 152, 147, 143, 115, 164, 160, 147, 150 (90%) Find the confidence interval of the median, indicated in parentheses, for the set of data.
-
The amount of milk obtained from a cow
-
Define each of the following terms: a. Assets-in-place; growth options; nonoperating assets b. Net operating working capital; operating capital; NOPAT; free cash flow c. Value of operations; horizon...
-
If you are certain you could earn a 20 percent return on an investment in some other companys stock, what would you do? Explain your reasoning. Assume you own stock in a publicly traded company and...
-
DeJohn Company, which began operations at the beginning of 2012, produces various products on a contract basis. Each contract generates a gross profit of $80,000. Some of DeJohns contracts provide...
-
Any assistance plz. 2. A stock price is currently selling at $100. Over each of the next two 6-month periods it is expected to go up by 10% or down by 10%. The risk-free interest rate for each of...
-
The adjusted trial balance for Speedy Courier as of December 31, 2013, follows. Required 1. Use the information in the adjusted trial balance to prepare (a) The income statement for the year ended...
-
The equilibrium constant for C + 1/2 O 2 CO 2 reaction at 100 kPa and 1600 K is K p . Use this information to find the equilibrium constant for the following reactions at 1600 K. (a) C + 1/2 O 2 CO...
-
At what temperature will oxygen be 15 percent disassociated at (a) 3 psia (b) 100 psia?
-
A diving pool that is 4 m deep and full of water has a viewing window on one of its vertical walls. Find the force on the following windows. The window is a circle, with a radius of 0.5 m, tangent to...
-
Mergers and acquisitions (M&A) are strategies that help companies to grow in size rapidly. However, some incredibly questionable M&A decisions were reported in the mining industry in 2012 and 2013,...
-
Why is a potential investor's perspective different than an existing investor's perspective?
-
WireOne makes mobile telephones. It currently has plans to manufacture the AZZ01 model. By redesigning the case at a cost of 95,000, it estimates that it can cut the repair cost of warranty work by...
-
In mid-2011, Sino-Forest Corporation was a company with timber operations in China, including tree plantation (holding of timber for appreciation and/or harvesting), log and wood products trading,...
-
List three financial statement users and a decision for each that may rest on financial statement analysis.
-
Cobb Company sells appliance service contracts to repair appliances for a two-year period. Cobb's past experience is that, of the total amount spent for repairs on service contracts, 40% is incurred...
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
A constant-volume tank contains a mixture of 120 g of methane (CH4) gas and 600 g of O2 at 25oC and 200 kPa. The contents of the tank are now ignited, and the methane gas burns completely. If the...
-
Reconsider Prob. 15-65. Using EES (or other) software, investigate the effect of the final temperature on the final pressure and the heat transfer for the combustion process. Let the final...
-
One lbmol of methane (CH4) undergoes complete combustion with stoichiometric amount of air in a rigid container. Initially, the air and methane are at 14.4 psia and 77oF. The products of combustion...
-
Does a mixed economy mean an economy where there is ? A . Existence of capitalism B . Privatization, liberalization and globalization C . Existence of both public and private sectors D . Growing...
-
Discussed in the chapter 13 and the article, "11 Weasel Words to Avoid in Conversation at All Costs" B weasel words are seen as unethical because organizations use them to
-
Consolidating entries ( fair value differs from book value ) Assume that on January 1 , 2 0 1 3 , an investor company acquired 1 0 0 % of the outstanding voting common stock of an investee company....

Study smarter with the SolutionInn App


