Methane (CH 4 ) is burned with 300 percent excess air in an adiabatic constant-volume container. Initially,
Question:
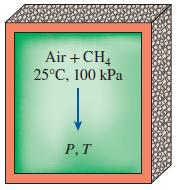
Methane (CH4) is burned with 300 percent excess air in an adiabatic constant-volume container. Initially, air and methane are at 1 atm and 25°C. Assuming complete combustion, determine the final pressure and temperature of the combustion products.
Transcribed Image Text:
Air +CH, 25°C, 100 kPa P,T
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
Methane is burned with 300 percent excess air adiabatically in a constant volume container The final ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Methane (CH4) is burned with 200 percent excess air in an adiabatic constant volume container. Initially, air and methane are at 1 atm and 25oC. Assuming complete combustion, determine the final...
-
Methane fuel (CH4) is burned with 15 percent excess air in a space-heating furnace. The pressure in the chimney is 1 atm. Presuming complete combustion, determine the temperature of the combustion...
-
Ethyl alcohol [C 2 H 5 OH(g)] is burned with 200 percent excess air in an adiabatic, constant-volume container. Initially, air and ethyl alcohol are at 100 kPa and 25C. Assuming complete combustion,...
-
This bar chart displays the demographics (age group and gender) of a Business Analysis class Business Analysts Students 23-33 3410 M lem How many male students are in the class? 65 80 130 50
-
In a survey, the Canadian Automobile Association (CAA) found that 6.1% of its members bought their cars at a used-car lot. If 15 CAA members are selected at random, what is the probability that 4 of...
-
What is the difference between an ordinary annuity and an annuity due? What type of annuity is shown below? How would you change it to the other type of annuity? Assume that you are nearing...
-
Michael Diaz Sporting Goods is considering the purchase of a machine that is used to cut material to make baseball gloves. The cost of the machine is \($265,000\). The machine has an estimated useful...
-
Casey Nelson is a divisional manager for Pigeon Company. His annual pay raises are largely determined by his divisions return on investment (ROI), which has been above 20% each of the last three...
-
Can you help me solve. Question 5 2 points Save Answer Affiliate A sells 7,000 units to Affiliate B per year. The marginal income tax rate for Affiliate A is 27 percent and the marginal income tax...
-
Write an ARM Assembly code subroutine to approximate the square root of an argument using the bisection method. Your code must approximate the square root of an integer between 0 and 2^(31) -1. Using...
-
A fuel at 25C is burned in a well-insulated steady flow combustion chamber with air that is also at 25C. Under what conditions will the adiabatic flame temperature of the combustion process be a...
-
A large railroad has experimented with burning powdered coal in a gas turbine combustor. Fifty percent excess air was introduced to the combustor at 1380 kPa and 127C while the powdered coal was...
-
Dorno Corporation reported expenses at the end of the current year as follows: $2,300 delivery expense, $5,500 advertising expense, $13,750 sales salaries expense, $1,250 sales supplies expense,...
-
How do the notions of going concern and gone concern affect ecological statements?
-
Why is monitoring and information disclosure critical for the success of emissions banking and trading?
-
What is an environmental intervention? How are environmental interventions and eco-efficiency related?
-
How are the limits on using eco-efficiency indicators related to accounting?
-
How important are environmental disclosures in the management discussion and analysis section of an annual report? Explain.
-
Ryan company paid salaries for the month amounting to $120,000. Of this amount, $30,000 was received by employees who had already been paid the $53,400 maximum amount of annual earnings taxable in...
-
Hardin Services Co. experienced the following events in 2016: 1. Provided services on account. 2. Collected cash for accounts receivable. 3. Attempted to collect an account and, when unsuccessful,...
-
Propane (C3H8) is burned with 150 percent theoretical air. The air-fuel mass ratio for this combustion process is (a) 5.3 (b) 10.5 (c) 15.7 (d) 23.4 (e) 39.3
-
One kmol of methane (CH4) is burned with an unknown amount of air during a combustion process. If the combustion is complete and there are 1 kmol of free O2 in the products, the air-fuel mass ratio...
-
An equimolar mixture of carbon dioxide and water vapor at 1 atm and 60oC enter a dehumidifying section where the entire water vapor is condensed and removed from the mixture, and the carbon dioxide...
-
Choose the authority bases that are most congruent with a teacher-directed theory of influence. Oreferent/expert O expert/legitimate Olegitimate/reward Oreward/referent
-
Duties of a principal to an agent include all except the duty to: Oshare financial benefits of the agency. O indemnify the agent for losses suffered while acting legally as directed by the principal....
-
In its earliest years, commercial radio often played A Live performances of music B Pre-recorded performances of music C Canned performances of music D) Composers writing music down in real time

Study smarter with the SolutionInn App


