On a summer day in New Orleans, Louisiana, the pressure is 1 atm: the temperature is 32C;
Question:
On a summer day in New Orleans, Louisiana, the pressure is 1 atm: the temperature is 32°C; and the relative humidity is 95 percent. This air is to be conditioned to 24°C and 60 percent relative humidity. Determine the amount of cooling, in kJ, required and water removed, in kg, per 1000 m3 of dry air processed at the entrance to the system.
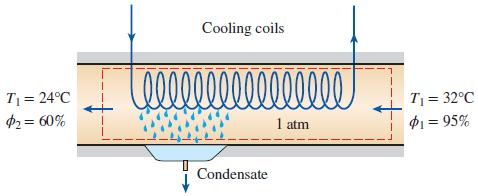
Cooling coils 00000000 T = 24°C $2= 60% T = 32°C P1 = 95% 1 atm Condensate
Step by Step Answer:
Air is cooled and dehumidified at constant pressure The amount of water removed from the air and the ...View the full answer



Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Related Video
In this video, the concept of pressure has been explained. The bottle is filled up with water, remove the pipe, and now you can see the clear flow of water from the holes due to liquid pressure.You can see that the second layer is holding the weight of the first layer and the third layer holding the weight of both upper layers, that’s why liquid flows out wider from the third as compared to the first two.At the first hole, water doesn’t roll along the walls; it flows out at an angle.At the second hole, you’ll see that the water flows out at a wider angle as compared to the first hole.At the third hole, we can see that the flow of water is much wider/greater than the other holes.This proves that the pressure at the bottom of the bottle is much more than the pressure at the top layers. P = ????gh
Students also viewed these Engineering questions
-
Reconsider Prob. 1482. How far will the temperature of the humid air have to be reduced to produce the desired dehumidification? Data From Reconsider Prob. 1482: On a summer day in New Orleans,...
-
During a summer day in El Paso, Texas, outdoor air is at 1 atm, 40C, and 20 percent relative humidity. Water at 20C is evaporated into this air to produce air at 25C and 80 percent relative humidity....
-
During a summer day in Phoenix, Arizona, the air is at 1 atm, 110oF, and 15 percent relative humidity. Water at 70oF is evaporated into this air to produce air at 75oF and 80 percent relative...
-
The following items were shown on the balance sheet of Herman Corporation on December 31, 2010: Stockholders Equity Paid-In Capital Capital Stock Common stock , $5 par value, 360,000 shares...
-
An observed frequency distribution is as follows: Number of successes 0 1 2 3 Frequency 89 133 52 26 a. Assuming a binomial distribution with n = 3 and p = 1/3, use the binomial probability...
-
A company reports the following income statement and balance sheet information for the current year: Net income $ 600,000 Interest expense 75,000 Average total assets 4,500,000 Determine the rate...
-
Yeast and Company has 5,000 shares of stock outstanding. The market value of Yeasts assets is \($800,000.\) The market value of its outstanding debt is \($200,000.\) The company issued 100 warrants...
-
Green Paper Delivery has decided to analyze the profitability of five new customers. It buys recycled paper at $ 20 per case and sells to retail customers at a list price of $ 26 per case. Data...
-
product lines. The company employs a standard cost accounting system for record-keeping purposes. At the beginning of 2012, the president of Sun Company presented the budget to the parent company and...
-
Power supplies for telecommunications applications may require high currents at low voltages. Design a buck converter that has an input voltage of 3.3 V and an output voltage of 1.2 V. The output...
-
Atmospheric air from the inside of an automobile enters the evaporator section of the air conditioner at 1 atm, 27C, and 50 percent relative humidity. The air returns to the automobile at 10C and 90...
-
Repeat Prob. 1479 for a total pressure of 88 kPa for air. Data From Repeat Prob. 1479: Air enters a 40-cm-diameter cooling section at 1 atm, 32C, and 70 percent relative humidity at 120 m/min. The...
-
A tank has two sides initially separated by a diaphragm. Side A contains 1 kg of water and side B contains 1.2 kg of air, both at 20C, 100 kPa. The diaphragm is now broken and the whole tank...
-
What is negative assurance within the context of an auditor's report? How does it differ from positive assurance?
-
Define a review of the financial statements of a nonpublic entity.
-
Discuss how both unions and firms can be better off if they move off the demand curve. Derive the contract curve.
-
What is the general reporting format on financial statements prepared in accordance with a comprehensive basis of accounting other than generally accepted accounting principles?
-
How does a financial projection differ from a financial forecast?
-
To what kinds of data sets can the empirical rule be applied?
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
An ideal-gas mixture consists of 2 kmol of N2 and 6 kmol of CO2. The mass fraction of CO2 in the mixture is (a) 0.175 (b) 0.250 (c) 0.500 (d) 0.750 (e) 0.875
-
An ideal-gas mixture consists of 2 kmol of N2 and 4 kmol of CO2. The apparent gas constant of the mixture is (a) 0.215kJ/kgK (b) 0.225kJ/kgK (c) 0.243kJ/kgK (d) 0.875kJ/kgK (e) 1.24kJ/kgK
-
A rigid tank is divided into two compartments by a partition. One compartment contains 3 kmol of N2 at 400 kPa and the other compartment contains 7 kmol of CO2 at 200 kPa. Now the partition is...
-
Eleanor is opening a store called "Everything is $10". As the name implies, all of the items sold at their shop will be priced at $10. They are estimating their variable cost per item they sells will...
-
Match the letter with the property of the improper integral. A The integral converges. B The integral diverges. Submit Answer Tries 0/8 3 L 6 dx x L 6 8 dx (x-3)4/5 3. S La Lo S 6 dx (3-x)4 6 34 dr...
-
Match the basic step in the recording process described by entering the appropriate statement number in the space provided. A. Analyze each transaction B. Enter each transaction in a journal C....

Study smarter with the SolutionInn App


