One kilogram of water vapor at 200 kPa fills the 1.1989-m 3 left chamber of a partitioned
Question:
One kilogram of water vapor at 200 kPa fills the 1.1989-m3 left chamber of a partitioned system shown in Fig. P3–37.
The right chamber has twice the volume of the left and is initially evacuated. Determine the pressure of the water after the partition has been removed and enough heat has been transferred so that the temperature of the water is 3°C.
Transcribed Image Text:
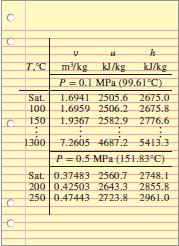
T.C m/kg K/kg P 0.1 MPa (99.61°C) KJ/kg Sat. T.6941 2505.6 2675.0 1.6959 2506.2 2675.8 100 150 1.9367 2582.9 2776.6 1300 7.2605 4687.2 5413.3 P=0.5 MPa (151.83°C) Sat. 0.37483 2560.7 2748.1 200 0.42503 2643.3 2855.8 250 0.47443 2723.8 2961.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
The initial specific volume is At the final state the water occ...View the full answer

Answered By

Wahome Michael
I am a CPA finalist and a graduate in Bachelor of commerce. I am a full time writer with 4 years experience in academic writing (essays, Thesis, dissertation and research). I am also a full time writer which assures you of my quality, deep knowledge of your task requirement and timeliness. Assign me your task and you shall have the best.
Thanks in advance
4.90+
63+ Reviews
132+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Tire City Inc. Mr. Abdullah, Chief Financial Officer (CFO) of Tire City Inc ('TCI') has a meeting with their bank later in the week. At the meeting he needs to present a request to the bank for...
-
During the current year, Rowan sells her entire interest in Concord Corporation common stock for $15,000. She is the sole shareholder, and originally organized the corporation several years ago by...
-
The radiator of a steam heating system has a volume of 20 L and is filled with superheated water vapor at 200 kPa and 200C. At this moment both the inlet and the exit valves to the radiator are...
-
If the current spot rate is 2.10 (HC/FC) and the government undertakes policies to depreciate the currency 16%, what is the new spot rate? Note: 2.10 * 1.16 is not the right formula. Based on this,...
-
A sporting goods manufacturing company wanted to compare the distance traveled by golf balls produced using each of four different designs. Ten balls of each design were manufactured and brought to...
-
How might you use IR spectroscopy to determine whether reaction between 2-cyclohexenone and lithium dimethylcopper gives the direct addition product or the conjugate product?
-
Describe a plan for selling a home.
-
Turney Company produces and sells automobile batteries, the heavy-duty HD-240. The 2014 sales forecast is as follows. Quarter HD-240 1 ......... 5,000 2 ......... 7,000 3 ......... 8,000 4 .............
-
The Gourmand Cooking School runs short cooking courses at its small campus. Management has identified two cost drivers it uses in its budgeting and performance reportsthe number of courses and the...
-
What are the advantages of investing in the common stock rather than the corporate bonds of a company? Compare the certainty of returns for a bond with those for a common stock . Draw a line graph to...
-
One kilogram of R-134a fills a 0.14-m 3 weighted pistoncylinder device at a temperature of 26.4C. The container is now heated until the temperature is 100C. Determine the final volume of the R-134a.
-
A rigid tank with a volume of 1.8 m 3 contains 40 kg of saturated liquid vapor mixture of water at 90C. Now the water is slowly heated. Determine the temperature at which the liquid in the tank is...
-
What is one possible value of x, if function f is undefined? f(x)= 5 2(x-2)-3(x-2)-2
-
"I'm going to work a hospital which is a not-for profit organization. Because there are no profits I will not able to apply any CVP analysis in my work." Do you agree with this statement? Why or why...
-
Are random arrivals at a shoe store at the local mall Poisson distributed? Suppose a mall employee researches this question by gathering data for arrivals during one-minute intervals on a weekday...
-
If 7.87 mol of ethane (C2H6) undergo combus- tion according to the unbalanced equation C2H6 + O2 CO2 + HO, how many moles of O2 is required? Answer in units of mol. 005 (part 2 of 3) 10.0 points How...
-
An electronics store has received a shipment of 30 table radios that have connections for an iPod or iPhone. Twelve of these have two slots (so they can accommodate both devices), and the other...
-
How's the weather? The following table presents the daily high temperatures for the city of Macon, Georgia, in degrees Farenheit, for the winter months of January and February in a recent year. 65 48...
-
A cube has 3 faces painted white, 2 faces painted red, and 1 face painted blue. What is the probability that a roll will result in a red or blue face?
-
The first national bank pays a 4% interest rate compound continuously. The effective annual rate paid by the bank is __________. a. 4.16% b. 4.20% c. 4.08% d. 4.12%
-
A 1-m3 tank contains 2.841 kg of steam at 0.6 MPa. Determine the temperature of the steam, using (a) The idealgas equation, (b) The van der Waals equation, and (c) The steam tables.
-
Reconsider Prob. 3-100. Solve the problem using EES (or other) software. Again using the EES, compare the temperature of water for the three cases at constant specific volume over the pressure range...
-
During a hot summer day at the beach when the air temperature is 30oC, someone claims the vapor pressure in the air to be 5.2 kPa. Is this claim reasonable?
-
Rent, depreciation, and salaries are examples of: A. current assets. B. current liabilities. C. owners equity. D. operating expenses.
-
https://youtu.be/yY96hTb8WgI Watch the following Video. This shows the bad of everyday design. There are some really simple ways to fix these bad design decisions. Using what we went through in...
-
Ho H = Calc ine and alternative hypotheses. Wins occur with all different frequency in the different post positions. x- =U At least one post position has a different frequency of wins than the...

Study smarter with the SolutionInn App


