Reconsider Prob. 1588. The automobile engine is to be converted to natural gas (methane, CH 4 )
Question:
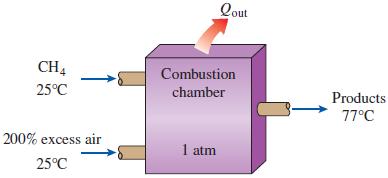
Reconsider Prob. 15–88. The automobile engine is to be converted to natural gas (methane, CH4) fuel. Assuming that all factors remain the same, what is the maximum work that can be produced by the modified engine, in kJ/kg fuel?
Data From Q#88:
n-Octane [C8H18(l)] is burned in an automobile engine with 200 percent excess air. Air enters this engine at 1 atm and 25°C. Liquid fuel at 25°C is mixed with this air before combustion. The exhaust products leave the exhaust system at 1 atm and 77°C. What is the maximum amount of work, in kJ/kg fuel, that can be produced by this engine? Take T0 = 25°C.
Transcribed Image Text:
Qout CH4 Combustion 25°C chamber Products 77°C 200% excess air 1 atm 25°C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
Methane is burned steadily with 200 percent excess air in a automobile engine The maximum amount of ...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
An automobile engine uses methyl alcohol (CH3OH) as fuel with 200 percent excess air. Air enters this engine at 1 atm and 25oC. Liquid fuel at 25oC is mixed with this air before combustion. The...
-
Liquid octane (C8H18) enters a steady-flow combustion chamber at 25C and 8 atm at a rate of 0.8 kg/min. It is burned with 200 percent excess air that is compressed and preheated to 500 K and 8 atm...
-
Liquid octane (C8H18) enters a steady-flow combustion chamber at 25oC and 8 atm at a rate of 0.8 kg/min. It is burned with 200 percent excess air that is compressed and preheated to 500 K and 8 atm...
-
XYZ purchased an equipment worth of $150,000 including insurance cost $30,000 and duty cost $20,000 on May 15, 2018. They have decided to capitalize the $35,000 installation charge instead of $20,000...
-
According to a market-share study, 13% of televisions in use are tuned to Hockey Night in Canada on Saturday night. Assume that an advertiser wants to verify that 13% market share value by conducting...
-
Wilson Wonderss bonds have 12 years remaining to maturity. Interest is paid annually, the bonds have a $1,000 par value, and the coupon interest rate is 10%. The bonds sell at a price of $850. What...
-
Following in random order are the four steps for evaluating a capital expenditure: Required: In the space provided, indicate a logical sequence of the steps for evaluating a capital expenditure. a....
-
Lenni Clothing Co. manufactures clothing in a small manufacturing facility. Manufacturing has 25 employees. Each employee presently provides 40 hours of productive labor per week. Information about a...
-
1.) The following statements about bonds are all correct, EXCEPT.... The par value of a bond is the amount of principal to be repaid at maturity Not all bonds have coupon payments Institutions and ...
-
Many financial managers and corporate officers have been criticized for (a) poor decisions, (b) lack of ethical behavior, (c) large salaries, (d) lucrative severance packages worth millions of...
-
n-Octane [C 8 H 18 (l)] is burned in an automobile engine with 200 percent excess air. Air enters this engine at 1 atm and 25C. Liquid fuel at 25C is mixed with this air before combustion. The...
-
A coal from Colorado which has an ultimate analysis (by mass) of 79.61 percent C, 4.66 percent H2, 4.76 percent O 2 , 1.83 percent N 2 , 0.52 percent S, and 8.62 percent ash (non combustibles) is...
-
A pure monatomic perfect gas with \(c_{p}=5 \Re / 2\) flows from one reservoir to another through a porous plug. The heat of transport of the gas through the plug is \(-\Re T / 2\). If the system is...
-
Portfolios are constructed to reduce risk for a given level of economic return. In this context, why would a company be interested in constructing an ecoefficiency portfolio?
-
Information management is the core of any environmental management system. Do you agree with this statement? How important is accounting information in an environmental management system? Outline...
-
Eco-control has five procedural steps.Think of an important corporate environmental intervention (e.g. emission of waste-water). With reference to this corporate environmental intervention, explain...
-
Why should business worry about the environment? How might ecological accounting help reduce such worries? In your answer consider the views of three different stakeholders.
-
Why might deep green environmentalists favour the presentation of corporate information in ecological accounts but reject disclosures in environmentally adjusted financial accounts?
-
State and describe at least four to five examples of milestones affecting the safety movement, from the 1800s until present. How has the role of organized labor played a significant role in the...
-
Explain why each of the following is either a private good or a public good: traffic lights, in line skates, a city park, a chicken salad sandwich, a tennis racket, national defense, a coastal...
-
In a combustion chamber, ethane (C2H6) is burned at a rate of 8 kg/h with air that enters the combustion chamber at a rate of 176 kg/h. Determine the percentage of excess air used during this process.
-
One kmol of ethane (C2H6) is burned with an unknown amount of air during a combustion process. An analysis of the combustion products reveals that the combustion is complete, and there are 3 kmol of...
-
Ethylene (C2H4) is burned with 175 percent theoretical air during a combustion process. Assuming complete combustion and a total pressure of 14.5 psia, determine (a) The air-fuel ratio (b) The...
-
Thu Jul 27 er ID and Password | California... ework Chapter 05 ales orders ales calls ervice calls verage order size anufacturing cost/unit ustomer costs: ctivity-Based Customer-Driven Costs uppose...
-
In a performance evaluation system, managers rank employees relative to one another. forced ranking the MBO process 360-degree feedback BARS
-
Identify the standard verb form of the following sentence.Every year my doctor _ _ _ _ _ me to take a flu shot, but I refuse. So far, I have never caught a virus.

Study smarter with the SolutionInn App


