The equation of state of a gas is given by where a and b are constants. Use
Question:
The equation of state of a gas is given by
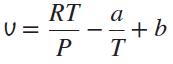
where a and b are constants. Use this equation of state to derive an equation for the Joule-Thomson coefficient inversion line.
Transcribed Image Text:
RT U = a + b T %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
The equation of state of a gas is given by An equation for the JouleThomson coefficient inv...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
The van der Waals equation of state, an approximate representation of the behavior of gases at high pressure, is given by Where a and b are constants having different values for different gases. (In...
-
The equation of state of a gas is given as (P + 10/ 2) = RuT, where the units of and P are m3/kmol and kPa, respectively. Now 0.5 kmol of this gas is expanded in a quasi-equilibrium manner from 2 to...
-
The equation of state of a gas is given as v-(P + 10/ v-2) = 5 RuT, where the units of v- and P are m3/kmol and kPa, respectively. Now 0.2 kmol of this gas is expanded in a quasi equilibrium manner...
-
What must be the beta of a portfolio with E( rP ) = 18%, if rf = 6% and E (rM) = 14%?
-
Find the regression equation; unless the problem suggests otherwise, let the first variable be the independent (x) variable. Caution: When finding predicted values, be sure to follow the prediction...
-
Consider the gas-solid equilibrium under the extreme assumption that the entropy of the solid may be neglected over the temperature range of interest. Let 0 be the cohesive energy of the solid, per...
-
Briefly explain the effects on interest recognized when the stated and market rates of interest are different.
-
Homepride Carpet Company manufactures carpets. Fiber is placed in process in the Spinning Department, where it is spun into yarn. The output of the Spinning Department is transferred to the Tufting...
-
As a part of the Audit, you are assessing internal control in the audit of the human resources and payroll cycle for the Sunshine Company, which is specialized in assembling computer systems from...
-
Maben Company was started on January 1, Year 1, and experienced the following events during its first year of operation: 1. Acquired $30,000 cash from the issue of common stock. 2. Borrowed $40,000...
-
What is the most general equation of state for which the Joule-Thomson coefficient is always zero?
-
What is the enthalpy departure?
-
What do the weights in a portfolio tell us?
-
A t-test is used when the number of degrees of freedom is unknown. In Exercises 9 and 10, determine whether the statement is true or false. If the statement is false, rewrite it as a true statement.
-
What are the similarities and differences in the form of accounts receivable and parable confirmation letters?
-
Johnson, CPA, was engaged to audit the financial statements of Horizon Incorporated, which has its own computer installation. While obtaining an understanding of internal control, Johnson found that...
-
Turpins COVID Labs, Inc. manages 26 laboratories in the United States. One laboratory located in Bonita Springs, Florida, began processing COVID-19 tests in early 2020 to address the need for rapid...
-
During an audit of a company that uses computers, Mr. Sure, the auditor, decided to process test data with some of the client's computer programs. The computer output matched his predesigned output....
-
The Harvard School of Public Health conducted a study of privately held firearm stock in the United States. In a representative household telephone survey of 2,770 adults, 26% reported that they own...
-
As economic conditions change, how do banks adjust their asset portfolio?
-
Why is the combined gas-steam cycle more efficient than either of the cycles operated alone?
-
The gas-turbine portion of a combined gas-steam power plant has a pressure ratio of 16. Air enters the compressor at 300 K at a rate of 14 kg/s and is heated to 1500 K in the combustion chamber. The...
-
Consider a combined gas-steam power plant that has a net power output of 450 MW. The pressure ratio of the gas-turbine cycle is 14. Air enters the compressor at 300 K and the turbine at 1400 K. The...
-
Chapter 13 Compare and contrast the similarities and differences of an air conditioning system with that of a refrigeration system? Provide a hand drawn diagram (no cutting and pasting of other...
-
1. (30 points) This gear train is similar to that in the assigned project; it has gears 1, 2, 3 and 4. The input is from the pulley (belt transmission) to the shaft Gear 1, n = 2400rpm and the input...
-
MET313: Applied Strength of Materials Week-5 Guided Practice Problem 1 (Columns) A round compression member with both ends pinned and made of AISI 1020 cold-drawn steel is to be used in a machine....

Study smarter with the SolutionInn App


