Consider an ideal vapor-compression cycle using R-134a and operating between pressures of 0.28 and 1.75 MPa. Determine
Question:
Consider an ideal vapor-compression cycle using R-134a and operating between pressures of 0.28 and 1.75 MPa. Determine the coefficient of performance for the cycle for application
(a) In a refrigerator
(b) In a heat pump.
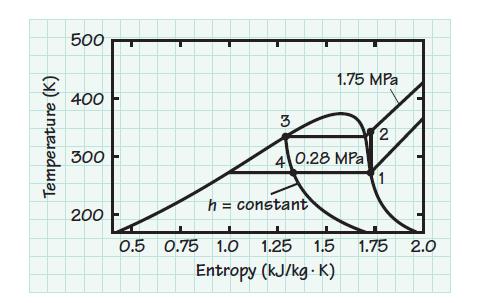
Transcribed Image Text:
Temperature (K) 500 400 300 200 0.5 3 0.75 1.0 h = constant 1.75 MPa 4 0.28 MPa 1.25 1.5 Entropy (kJ/kg. K) 2 1 1.75 2.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
To determine the coefficient of performance COP for the ideal vaporcompression cycle using R134a we ...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Determine the heat pump coefficient of performance for an ideal vapor compression refrigeration cycle operating between 0.653 and 1.342 MPa. The working fluid is R-22. Compare your result with that...
-
A heat pump that operates on the ideal vapor compression cycle with refrigerant-134a is used to heat a house and maintain it at 75F by using underground water at 50F as the heat source. The house is...
-
A refrigerator uses R-134a as the working fluid and operates on an ideal vapor compression refrigeration cycle between 0.15 MPa and 1 MPa. A temperature difference of 5C is maintained for effective...
-
- The first stream is $450 per year for 9 years and begins 5 years from today - The second stream begins 7 years from today with the first cash flow being $400, and with each successive cash flow...
-
The Alaska Airlines balance sheet dated December 31, 2011, included the following ($ in millions): Property and equipment Aircraft and other flight equipment ........$4,041.8 Other property and...
-
In Exercises, use limits to compute f(x). f(x)=xx
-
Read Joseph McCafferty's article entitled "Misgivings" at CFO.com (January 2007); https://www.cfo.com/accounting-tax/2007/01/misgivings/ (accessed 12/10/19). a. Discuss your thoughts about...
-
For more than 75 years, New England Foundry, Inc., has manufactured wood stoves for home use. In recent years, with increasing energy prices, George Mathison, president of New England Foundry, has...
-
Using Essential Bundle Food Data construct a two-way table Name of Store Day of Week Raleys Monday Raleys Tuesday Raleys Tuesday Walmart Friday Target Monday Target Wednesday Walmart Saturday Raleys...
-
MVP, a manufacturing firm with no debt outstanding and a market value of $100 million, is considering borrowing $40 million and buying back stock. Assuming that the interest rate on the debt is 9%...
-
Repeat Problem 9.117 using EES. Modify your EES program to find the coefficient of performance between pressures of 0.28 and 1.5 MPa. Plot Ts diagrams for the original cycle conditions and for the...
-
Heat leaks from the air in a kitchen through the walls of a refrigerator into the refrigerated cold space at a rate of 1.43 kW. Determine the electrical power required to maintain a steady...
-
Outline three criticisms of traditional budgeting and a related proposal for change.
-
Question-1 For the assigned system listed in Table-1 (system figures given in the annex-1) (70P); 1. Find the equation of motion using any of the following methods; a. Using Newton's Method, www -24-...
-
A new island is established and the citizens and their elected government plan to set up economic activity based on the current resources on the island and known technologies, they have determined...
-
A state government levies an estate tax, which taxes the survivors of a wealthy person if they inherit more than $2 million of cash or property from the deceased individual. Which market failure (if...
-
Use the compliance method to obtain the stress intensity factor for the DCB specimen shown below. The spring associated with the loading is assumed to be linear (with stiffness k). The specimen...
-
How do you determine the purpose and context of a memorandum, and what key elements should be included to ensure the memo effectively communicates its intended message?
-
For the Adams-Bashforth and Adams-Moulton methods of order four, a. Show that if f = 0, then F(ti , h,wi+1, . . . ,wi+1m) = 0. b. Show that if f satisfies a Lipschitz condition with constant L, then...
-
Frontland Advertising creates, plans, and handles advertising campaigns in a three-state area. Recently, Frontland had to replace an inexperienced office worker in charge of bookkeeping because of...
-
A spring-loaded pistoncylinder device contains 5 kg of helium as the system, as shown in Fig. P477. This system is heated from 100 kPa and 20C to 800 kPa and 160C. Determine the heat transferred to...
-
A balloon initially contains 40 m 3 of helium gas at atmospheric conditions of 100 kPa and 17C. The balloon is connected by a valve to a large reservoir that supplies helium gas at 125 kPa and 25C....
-
Water is boiled at 100C electrically by a 3-kW resistance wire. Determine the rate of evaporation of water. Steam Water 100C
-
Hi this is a question about business finance, thank you!
-
You interviewed a new client and these were the facts provided. Provide them with a brief explanation of tax reason for each Their home mortgage interest is $ 1 2 0 0 , real estate taxes $ 8 , 2 0 0...
-
Part A Capital Budgeting Background: Electro Motors (Electro) is considering a new project to produce electric vehicles for the Australian domestic market and international markets. It has identified...

Study smarter with the SolutionInn App


