Consider the equilibrium dissociation of carbon dioxide CO 2 CO + 1/2 O 2 . At
Question:
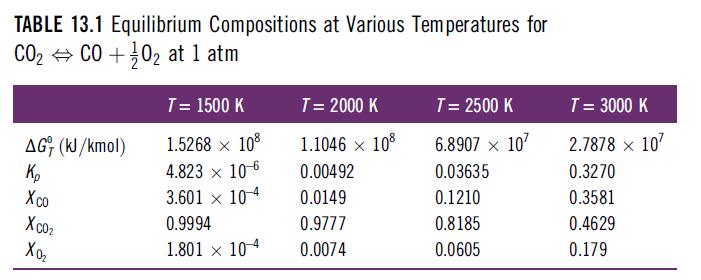
Consider the equilibrium dissociation of carbon dioxide CO2 ⇔ CO + 1/2 O2. At 2500 K, the equilibrium constant is 0.03635. Calculate the enthalpy of reaction for this reaction at 2500 K and use this to estimate the equilibrium constant for a temperature of 3000 K. Compare your estimated value with the exact value from Table 13.1 in Example 13.2 and discuss.
Table 13.1 in Example 13.2
Transcribed Image Text:
TABLE 13.1 Equilibrium Compositions at Various Temperatures for CO₂ CO +02 at 1 atm AGT (kJ/kmol) Кр Xco Xcoz Xo₂ T = 1500 K 1.5268 x 108 4.823 x 10-6 3.601 104 0.9994 1.801 x 104 T = 2000 K 1.1046 × 108 0.00492 0.0149 0.9777 0.0074 T = 2500 K 6.8907 x 107 0.03635 0.1210 0.8185 0.0605 T = 3000 K 2.7878 x 10² 0.3270 0.3581 0.4629 0.179
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
To calculate the enthalpy of reaction for the equilibrium dissociation of carbon dioxide at 2500 K w...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
The data in the table concern the lactonization of hydroxyvaleric acid at 25oC. They give the concentration C(t)of this acid in moles per liter after minutes. (a) Find the average rate of reaction...
-
Consider the reaction 2 CO2 2 CO + O2 obtained after heating 1 kmol CO2 to 3000 K. Find the equilibrium constant from the shift in Gibbs function and verify its value with the entry in Table A.11....
-
This question draws from the tables in the previous question. Let us try to get an idea of what it would cost an American family at todays prices to purchase the bundle consumed by an average Swedish...
-
Rice and Flower were partners sharing profit and loss equally. Statement of Financial Position as at 31 December 2020. Non current Assets Premises Machinery Vehicles Fittings Current Assets Inventory...
-
Form groups of three to six students each. Each student should pick two companies, preferably from different industries. Find the appropriate data and compute the market-to-book ratio and the ROE for...
-
Question 3 of 3 Additional information: 1. Net income for 2022 was $104,300. 2. Depreciation expense was $32,300. 3. Cash dividends of $46,300 were declared and paid. 4. Bonds payable with a carrying...
-
Ambassador Watch Company budgeted the following costs for anticipated production for April 2002: Prepare a factory overhead cost budget, separating variable and fixed costs. Assume that all indirect...
-
Henderson Company had an unadjusted cash balance of $7,215 as of May 31. The companys bank statement, also dated May 31, included a $68 NSF check written by one of Hendersons customers. There were...
-
Sandra is an ambitious producer with the Kramer Group. She is currently working with the Hopper Company, a small manufacturer who wants to explore other insurers but needs help assessing exactly what...
-
Gemini, Inc., an all-equity firm, is considering an investment of $1.4 million that will be depreciated according to the straight-line method over its four-year life. The project is expected to...
-
Ammonia is used as a refrigerant in large-scale refrigeration systems. Using data for ammonia (NH 3 ) from the NIST resources, verify that the condition for phase equilibrium (Eqs.13.35) is met. Use...
-
1. Consolidation workpaper entries normally: a Are posted to the general ledger accounts of one or more of the affiliates b Are posted to the general ledger accounts only when the financial statement...
-
Describe the role that information technology can play in enhancing supply chain security. Focus the discussion on the following technologies: RFID, shipment tracking and tracing, supply chain...
-
Mason Company has two manufacturing departments Machining and Assembly. The company considers all of its manufacturing overhead costs to be fixed costs. It provided the following estimates at the...
-
Construct the forecasting cash flow statement (Excel) based on Exhibit 7. Note: ignoring the interests, you should be able to calculate the depreciation through the CAPEX and Net PPE to determine the...
-
Northwood Company manufactures basketballs. The company has a ball that sells for $ 2 5 . At present, the ball is manufactured in a small plant that relies heavily on direct labor workers. Thus,...
-
Emmett and Sierra formed a partnership dividing income as follows:Annual salary allowance to Emmett of $ 3 8 , 5 0 0 Interest of 8 % on each partner's capital balance on January 1 Any remaining net...
-
Malone Company makes and sells products with variable costs of $ 2 4 each. Malone incurs annual fixed costs of $ 4 2 8 , 4 0 0 . The current sales price is $ 8 7 . Note: The requirements of this...
-
Suppose A = PtLU, where P is a permutation matrix, L is a lower-triangular matrix with ones on the diagonal, and U is an upper-triangular matrix. a. Count the number of operations needed to compute...
-
How does the organizational structure of an MNC influence its strategy implementation?
-
An engineer is studying the mileage performance characteristics of five types of gasoline additives. In the road test he wishes to use cars as blocks; however, because of a time constraint, he must...
-
Construct a set of orthogonal contrasts for the data in Problem 4-27. Compute the sum of squares for each contrast. Problem 4-27. An engineer is studying the mileage performance characteristics of...
-
Seven different hardwood concentrations are being studied to determine their effect on the strength of the paper produced. However, the pilot plant can only produce three runs each day. As days may...
-
II. Roger's Manufacturing Company is selling its classic Rolls-Royce Phantom in mint condition for $200,000 at a public auction. The car was originally bought for $60,000 and it has been always kept...
-
The RLX Company just paid a dividend of $3.05 per share on its stock. The dividends are expected to grow at a constant rate of 5.5 percent per year, indefinitely. Assume investors require a return of...
-
Describe the topic you have chosen to study and explain what kinds of data you will collect to present to your government representative. Describe two specific recommendations you would make to...

Study smarter with the SolutionInn App


