3 kg of ammonia at its critical state is placed in a constant-volume container, which is then...
Question:
3 kg of ammonia at its critical state is placed in a constant-volume container, which is then cooled until the temperature is 300 K. How a much energy was transferred as heat from the ammonia in this process, and what tank volume is occupied by liquid at the end of the process? Use the tables in Appendix A.7 for data.
Data From Appendix A.7
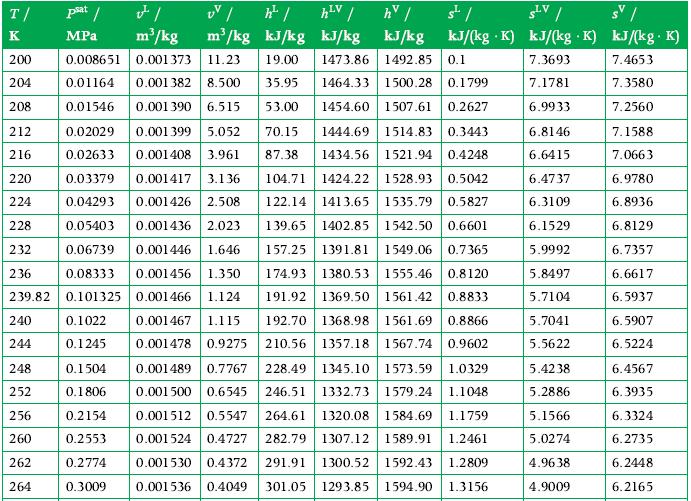
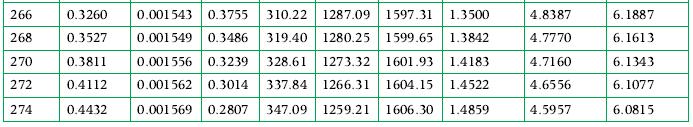
Transcribed Image Text:
T/ K UV / ¹² / of / m³/kg MPa m³/kg kJ/kg 200 0.008651 0.001373 11.23 19.00 204 0.01164 0.001 382 8.500 35.95 208 0.01546 0.001 390 6.515 53.00 212 0.02029 0.001 399 5.052 70.15 1444.69 1514.83 0.3443 216 0.02633 0.001408 3.961 87.38 1434.56 1521.94 0.4248 220 224 228 232 0.03379 0.001417 3.136 104.71 1424.22 1528.93 0.5042 0.04293 0.001426 2.508 122.14 1413.65 1535.79 0.5827 0.05403 0.001436 2.023 139.65 1402.85 1542.50 0.6601 0.06739 0.001446 1.646 157.25 1391.81 1549.06 0.7365 236 0.08333 0.001456 1.350 174.93 1380.53 1555.46 0.8120 239.82 0.101325 0.001466 1.124 191.92 1369.50 1561.42 0.8833 240 0.1022 0.001467 1.115 192.70 1368.98 1561.69 0.8866 0.1245 210.56 1357.18 1567.74 0.9602 244 0.001478 0.9275 0.001489 0.7767 228.49 1345.10 1573.59 1.0329 0.001500 0.6545 246.51 1332.73 1579.24 1.1048 0.001512 0.5547 264.61 1320.08 1584.69 1.1759 0.001524 0.4727 282.79 1307.12 1589.91 1.2461 0.001530 0.4372 291.91 1300.52 1592.43 1.2809 0.001536 0.4049 301.05 1293.85 1594.90 1.3156 248 252 256 260 262 264 0.1504 0.1806 hv / "/ sv/ kJ/kg kJ/(kg K) kJ/(kg K) kJ/(kg. K) 7.3693 7.4653 7.1781 7.3580 6.9933 7.2560 6.8146 7.1588 6.6415 7.0663 6.4737 6.3109 6.1529 5.9992 5.8497 5.7104 5.7041 5.5622 0.2154 0.2553 0.2774 0.3009 hLV | kJ/kg 1473.86 1492.85 0.1 1464.33 1500.28 0.1799 1454.60 1507.61 0.2627 SLV 5.4238 5.2886 5.1566 5.0274 4.9638 4.9009 6.9780 6.8936 6.8129 6.7357 6.6617 6.5937 6.5907 6.5224 6.4567 6.3935 6.3324 6.2735 6.2448 6.2165
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The energy balance for the iso...View the full answer

Answered By

Swarnim Raut
I tutored few juniors of mine during my Mechanical Engineering and my high school. I have the ability to explain difficult concepts into simple manner. My love for Mechanical Engineering has allowed me to get in depth knowledge of the subject. I like to learn and share it with others.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
Please need HELP ASAP! FOLLOW THE INSTRUCTIONS!! Problem 3: Presented below is information related to Macaw Corp., for the year 2019. Net sales Cost of goods sold Selling expenses Administrative...
-
Propane is in a 5 m 3 container as a saturated vapor at 300 K. The sealed tank is then cooled to 260 K by fresh snow. What are the pressure (Pa) and liquid volume in this 260 K state, and how much...
-
One kilogram of R134a is heated in a constant volume container from the critical point to 450 K. How much energy is transferred as heat to the fluid? Use the diagrams or data in Appendix A.4. Data...
-
Design a plan to research and select a new or used automobile.
-
The pre-closing, year-end trial balance for a capital projects fund of the city of Rochester as of December 31, 2019, follows: Required 1. Prepare closing entries as of December 31, 2019. 2. Prepare...
-
Iodine-131 has a half-life of 8.0 days. How many days will it take for 174 g of 131 I to decay to 83 g of 131 I?
-
Refer to Exercise 5-6 and prepare journal entries to record each of the merchandising transactions assuming that the periodic inventory system is used by both the buyer and the seller, Exercise 5-6...
-
Palomar Chemical Company produces three products: ethylene, butane, and ester. Each of these products has high demand in the market, and Palomar Chemical is able to sell as much as it can produce of...
-
Here are the hottest recorded temperatures (in F) for each of sixt Temperatures (in F) 110 105 96 105 97 93 104 99 106 112 103 101 102 94 44 99 94 Send data to calculator (a) Complete the grouped...
-
Kayak Company budgeted the following cash receipts (excluding cash receipts from loans received) and cash payments (excluding cash payments for loan principal and interest payments) for the first...
-
A free-piston engine under development consists of a small piston in a cylinder. Each end of the cylinder behaves like a two-stroke engine, in which the following sequence of processes occurs...
-
Determine the equivalents of 1 m, 1 kg, 1 N, 1 J, and 1 W in a Systme Unis in which both k N and k G are unity, the speed of light is unity, and the second is the only primary unit. Choose a suitable...
-
We have seen that determinants can be used to solve linear equations, give areas of triangles in rectangular coordinates, and determine equations of lines. Not impressed with these applications?...
-
If you were appointed CM consultant how would you work with CEO and middle management to address the challenges & issues coming out of the adoption & move to AI at the company, Also, develop a step...
-
The goal is to link theory in HRM practices or programs. Your research is centered on employee engagement and organizational branding. Imagine that you are a new HRM manager providing a summary paper...
-
Can you explain to me and the rest of the class the importance of reviewing for routine messages? For instance, what considerations should you make and what are your primary goals? Please support...
-
During periods of internal business growth, what kind of employee training would be a good place to focus? What aspect you should do during this time period?
-
Are there different types of research used to identify market segments? If there are, can you provide three types of research that are used to identify market segments? Explain with the help of...
-
An energy-storage network consists of series-connected 16-mH and 14-mH inductors in parallel with a series connected 24-mH and 36-mH inductors. Calculate the equivalent inductance.
-
What is your opinion of advertising awards, such as the Cannes Lions, that are based solely on creativity? If you were a marketer looking for an agency, would you take these creative awards into...
-
Michael Phelps won a record-setting 8 gold medals at the 2008 Beijing Olympics. Now imagine if Phelps had competed in a pool filled with pancake syrup. Would you expect his race times to increase,...
-
The fuel tank on a sport-utility wagon holds 14 gal of gasoline. How much heavier is the automobile when the tank is full compared to when it is empty?
-
The pressure at the bottom of an 18-ft-deep storage tank for gasoline is how much greater than at the top? Express your answer in the units of psi.
-
In developing a budget, which of the following should be considered? I. Balance sheet II. Chart of accounts III. Expenses from the current/past year IV. Purchasing contracts V. Physician compensation...
-
JOIICS SUIT SHOPSales BudgetQuarter 1Quarter 2Quarter 3Expected Sales Units150175Sales Price per Unit$250250$250otal Sales Revenue37,500$ JOIICS SUIT SHOPSales BudgetQuarter 1Quarter 2Quarter...
-
Which of the following statements is true concerning foreign income tax paid? Question 39Select one: a. If a taxpayer takes a subtraction on their Oregon return, then the subtraction is included as...

Study smarter with the SolutionInn App


