A laboratory analysis shows that CO 2 becomes 10 % dissociated into CO and O 2 at
Question:
A laboratory analysis shows that CO2 becomes 10 % dissociated into CO and O2 at 2390 K if the total pressure is 1 atm. Obtain the equilibrium constant from this information and compare it to the value given in Table D.1.
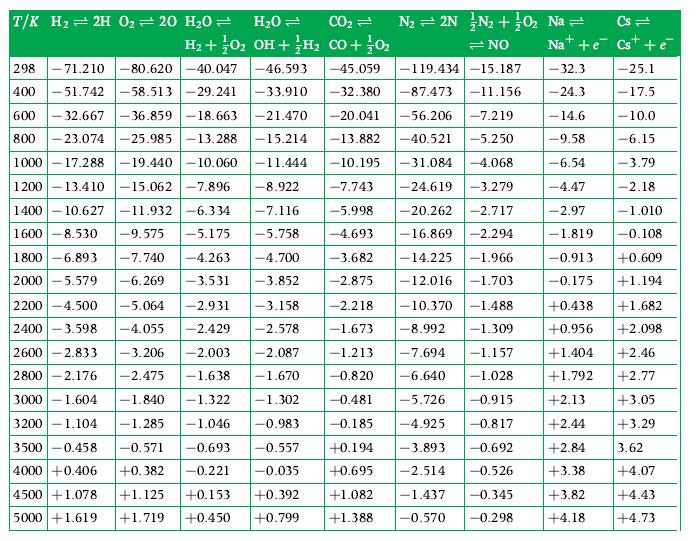
Transcribed Image Text:
T/K H₂2H 0₂ = 20 H₂0 = H₂O = CO₂ = N₂ = 2N N₂ + 0₂ Na = Cs= H₂ + O2 OH + H₂ CO + 0₂ =NO Nat+e Cs + é -45.059 -32.3 -32.380 -87.473 -11.156 -24.3 -20.041 -56.206 -7.219 - 14.6 -13.882 -40.521 -5.250 -9.58 -10.195 -31.084 -4.068 -6.54 298 -71.210 -80.620-40.047 -46.593 400 -51.742-58.513 -29.241 -33.910 600 -32.667 -36.859-18.663 -21.470 800-23.074 -25.985 -13.288 -15.214 1000-17.288 -19.440 -10.060 -11.444 1200-13.410 -15.062 -7.896 -8.922 1400-10.627 -11.932-6.334 -7.116 1600 -8.530 -9.575 -5.175 -5.758 1800 -6.893 2000 -5.579 -4.47 -2.97 -7.743 -24.619 -3.279 -5.998 -20.262 -2.717 -4.693 -16.869 -2.294 -3.682 -14.225 -1.966 -2.875 -12.016 -1.703 -1.819 -7.740 -4.263 -4.700 -3.852 -6.269 -3.531 2200 -4.500 -5.064 -2.931 2400 -3.598 -4.055 -2.429 2600 -2.833 -3.206 -2.003 2800 -2.176 -2.475 -1.638 3000 -1.604 -1.840 -1.322 -2.218 -10.370 -1.488 -1.673 -8.992 -1.309 -7.694 -1.157 -6.640 -1.028 -5.726 -0.915 -4.925 -0.817 -3.893 -0.692 -2.514 -0.526 -0.345 -0.298 -3.158 -2.578 -2.087 -1.213 -1.670 -0.820 - 1.302 -0.481 -0.185 +0.194 +0.695 3200-1.104 -1.285 -1.046 -0.983 3500 -0.458 -0.571 -0.693 -0.557 4000 +0.406 +0.382 -0.221 -0.035 4500 +1.078 +1.125 +0.153 5000 +1.619 +1.719 +0.450 +0.392 +0.799 -119.434-15.187 +1.082 -1.437 +1.388. -0.570 -25.1 -17.5 -10.0 -6.15 -3.79 -2.18 -1.010 -0.108 -0.913 +0.609 -0.175 +1.194 +1.682 +2.098 +2.13 +2.44 +2.84 +3.38 +0.438 +0.956 +1.404 +2.46 +1.792 +2.77 +3.05 +3.29 3.62 +4.07 +3.82 +4.43 +4.18 +4.73
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
The reaction can be written as CO CO 050 As the laboratory analysi...View the full answer

Answered By

Lilian Nyambura
Hi, am Lilian Nyambura, With extensive experience in the writing industry, I am the best fit for your writing projects. I am currently pursuing a B.A. in Business Administration. With over 5 years of experience, I can comfortably say I am good in article writing, editing and proofreading, academic writing, resumes and cover letters. I have good command over English grammar, English Basic Skills, English Spelling, English Vocabulary, U.S. English Sentence Structure, U.K. or U.S. English Punctuation and other grammar related topics. Let me help you with all your essays, assignments, projects, dissertations, online exams and other related tasks. Quality is my goal.
4.80+
378+ Reviews
750+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
A sample of O2 is collected over water in a 5.00 L container at 20C. If the total pressure is 688 torr, how many moles of O2 are collected?
-
In a gas mixture of O2 and N2, the total pressure is 2.66 atm and the partial pressure of O2 is 888 torr. What is the partial pressure of N2?
-
A sample of CO is collected over water in a 25.00 L container at 5C. If the total pressure is 0.112 atm, how many moles of CO are collected?
-
Ebrahim Patel is a wholesaler who uses the periodic inventory system to account for inventory. Transactions for February: 1 Bought inventory from Rich Traders for R5 000 on credit. 2 Sold inventory...
-
The town of Musicville has two residents: Bach and Mozart. The town currently fund sits free outdoor concert series solely from the individual contributions of these residents. Each of the two...
-
Rewrite the following pseudocode segment using a loop structure in the specified languages: a. C, C++, Java, or C# b. Python c. Ruby Assume all variables are integer type. Discuss which language, for...
-
Why is it difficult for many nonprofits to measure and evaluate their impact? Can you think of any nontraditional ways of assessing the effectiveness of an organization?
-
Friedas Fizz brews specialty soft drinks, including root beer and other flavors. Its vision is To proudly produce and sell extraordinarily smooth, rich, and delicious soft drinks to satisfy kids of...
-
Check my wo Problem 13-4 Discounted Payback (LG13-2) Compute the discounted payback statistic for Project C if the appropriate cost of capital is 8 percent and the maximum allowable discounted...
-
Clear Lake Nursing Home defines outputs as a patient day. Its present volume is 26,000 patient days. The average cost per day is $90,00. Present revenues and costs are presented below: Clear Lake...
-
A turboprop engine has the following characteristics: The engine propels an airplane at 640 km/hr at an altitude of 7600 m (?35 ?C, 38 kPa). Analyze the thermodynamic cycle on a per kg basis, making...
-
Propane (C 3 H 8 ) and oxygen in stoichiometric proportions react in a steady flow water-cooled burner. The reactants enter the burner at 305 K and leave the burner at 2 atm and emerge at 730 K. The...
-
Outline the main cultural and organizational problems associated with change that are described in the case.
-
A metal plate of aluminum is exposed an oxygen rich atmosphere on one side and an oxygen deficient atmosphere on the other side, at 500K. Assuming steady-state diffusion has been achieved, calculate...
-
Evaluate 7m+3mn when m = 8 and n = 14
-
Nowater, Inc. completed Job No. H12 during the year. The job cost sheet listed the following: Direct materials Direct labor Manufacturing overhead applied Units produced Units sold $110,000 $60,000...
-
How are advertising and public relations different? A trend within the ad industry is to merge Madison and Vine. What does this mean and how has it changed the face of marketing? We discussed...
-
Find lim x-2x-35 x-5 x+3x-10
-
Pujah Srinivasan is the controller for HHT Industries. She has been asked to explain the payroll accounts on the financial statements for the preceding month. What information will she find about...
-
Sheldon and Leonard had a million-dollar idea. In order to make it happen, they have to do special research first. Only Kripke can help them in this matter. But Kripke is known to be the first-class...
-
Spreadsheet for standard deviation. Lets create a spreadsheet to compute the mean and standard deviation of a column of numbers in two different ways. The spreadsheet here is a template for this...
-
Use Table 4-1 for this exercise. Suppose that the mileage at which 10000 sets of automobile brakes had been 80% worn through was recorded. The average was 62700, and the standard deviation was 10 400...
-
Use the NORMDIST spreadsheet function to answer these questions about the brakes described in Exercise 4-C: (a) What fraction of brakes is expected to be 80% worn in less than 45 800 miles? (b) What...
-
The payroll disbursements were for "two persons" named Coitty and Coitty with the same first name and address. The interesting observation is that Coitty is dated February 28, 2019 or after while...
-
What was the market capitalization of tesla in 2 0 2 2
-
Cundo debe presentarse las plataformas digitales la declaracin informativa en el portal del SAT, respecto a todos sus clientes que hacen uso de ella para la enajenacin de bienes, prestacin de...

Study smarter with the SolutionInn App


